A menu of options: Extended release versions of opioid use disorder medications may increase uptake and decrease stigma
Despite strong evidence of the effectiveness of FDA-approved medications in reducing opioid use and overdose, only a subset of individuals with opioid use disorder who seek treatment are prescribed such medications. Long-acting injectable and implantable forms of buprenorphine (the oral formulation of which is also known by the common brand names Subutex or Suboxone when combined with naloxone) are emerging as new options. Will this innovation make medications more attractive among individuals with opioid use disorder?
WHAT PROBLEM DOES THIS STUDY ADDRESS?
There has been an underutilization of life saving opioid agonist medications like buprenorphine even though there is strong evidence these medications decrease mortality, reduce illicit opioid use, and increase retention in treatment. In fact, only one-third of individuals treated for opioid use disorder receive medication treatment. Some of this underutilization is related to stigma, such as the belief that opioid agonist medication is “substituting one addiction for another” and the potential for the medication to be diverted and misused. Long-acting versions of opioid use disorder medications have emerged, including extended-release naltrexone in a monthly injectable, weekly and monthly injectables of buprenorphine, and buprenorphine implants lasting six months. Long-acting forms of buprenorphine have been shown to be viable alternatives to oral formulations, though factors impacting adoption and patient preferences are unclear. There is much interest among clinicians for the potential of these medications to increase compliance. However, it is important to examine the preferences of the people with opioid use disorder who would take these medications.
A prior study showed preferences among people in the United Kingdom for long-acting medications for opioid use disorder were likely influenced by a wide range of factors but were generally positive. A study in the United States among a treatment-seeking population found that about half preferred short-acting medications – for example, oral buprenorphine formulations taken daily (e.g., Suboxone) – and the other half preferred some version of long-acting medications for opioid use disorder (injection or implant). In this study, the authors sought to better understand the preferences for short-acting versus long-acting medications among individuals with opioid use disorder in the United States, some of whom were currently receiving treatment and some of whom were not. With only 20% of individuals with opioid use disorder receiving any treatment and only a third of these treated individuals receiving medication treatment, perspectives from those who are not currently seeking treatment may inform treatment modalities involving medications that are acceptable and attractive, thus increasing utilization and decreasing stigma.
HOW WAS THIS STUDY CONDUCTED?
This was a qualitative study with 40 participants (24 men and 16 females, aged 23-65 years) who were purposefully enrolled to reside in and represent different regions of the U.S. and to have varying opioid use disorder treatment experiences. Study participants were evaluated on their opioid use disorder treatment preferences through a semi-structured interview, with a focus on preferences for short-acting or long-acting forms of opioid use disorder medications. Responses were audio recorded, transcribed, and analyzed for themes.
This research was conducted in the United States between February and March of 2018. Study participants were purposefully enrolled to represent geographically diverse regions and varying opioid use disorder treatment experiences through two methods: Craigslist advertisements and flyers in addiction treatment programs. Eligible participants were 18 years of age or older and scored at least a two on either the heroin or the opioid pain reliever sections of the Tobacco, Alcohol, Prescription Medications, and Other Substances (TAPS) Tool, suggestive of having an opioid use disorder.
Among the 40 study participants, most were predominantly White (90%) and just over half were male (60%), lived in highly populated areas of the United States (80%), had a high school degree or higher education (90%), and had a stable housing situation (97.5%). There was a range of reported last substance use, as 25% of study participants had used in the past week and 37.5% had not used in the past six months. Thirty-six (90%) had a history of opioid use disorder treatment, 24 (60%) had a history of opioid use disorder treatment using medications, and 10 (25%) were currently on some form of opioid use disorder medication. Specifically, eight participants had been prescribed methadone only, seven participants had been prescribed buprenorphine-naloxone (also known as Suboxone) only, two participants had been prescribed naltrexone only, seven participants had been prescribed two different types of medications for opioid use disorder, and one participant had been prescribed all three types. Five participants had previous experience with injectable and implantable forms of opioid use disorder medication, primarily extended-release naltrexone (60%).
Study participants were interviewed via telephone, each lasting between 45-60 minutes. For those study participants who had access to an email account, an information tool (see adapted graphic below) was provided before the interview about different forms of opioid use disorder medications. Interviews were conducted using an interview guide, a tool used in qualitative research to direct the conversation toward the topics and issues relevant to the study. The guide was developed based on both a scientific literature review and input from experts, focusing on participants’ history of opioid use, previous experiences with opioid use disorder treatment, and knowledge and preferences for long-acting pharmacotherapies.
Interviews were audio recorded and transcribed verbatim, and the qualitative data from the interviews was coded with statistical software and analyzed for themes. Researchers framed the findings of the interviews using a conceptual model of patients’ lived experience with medicine (PLEM) which is comprised of three domains: medication-related beliefs, medication-related burden, and medication-taking practices.
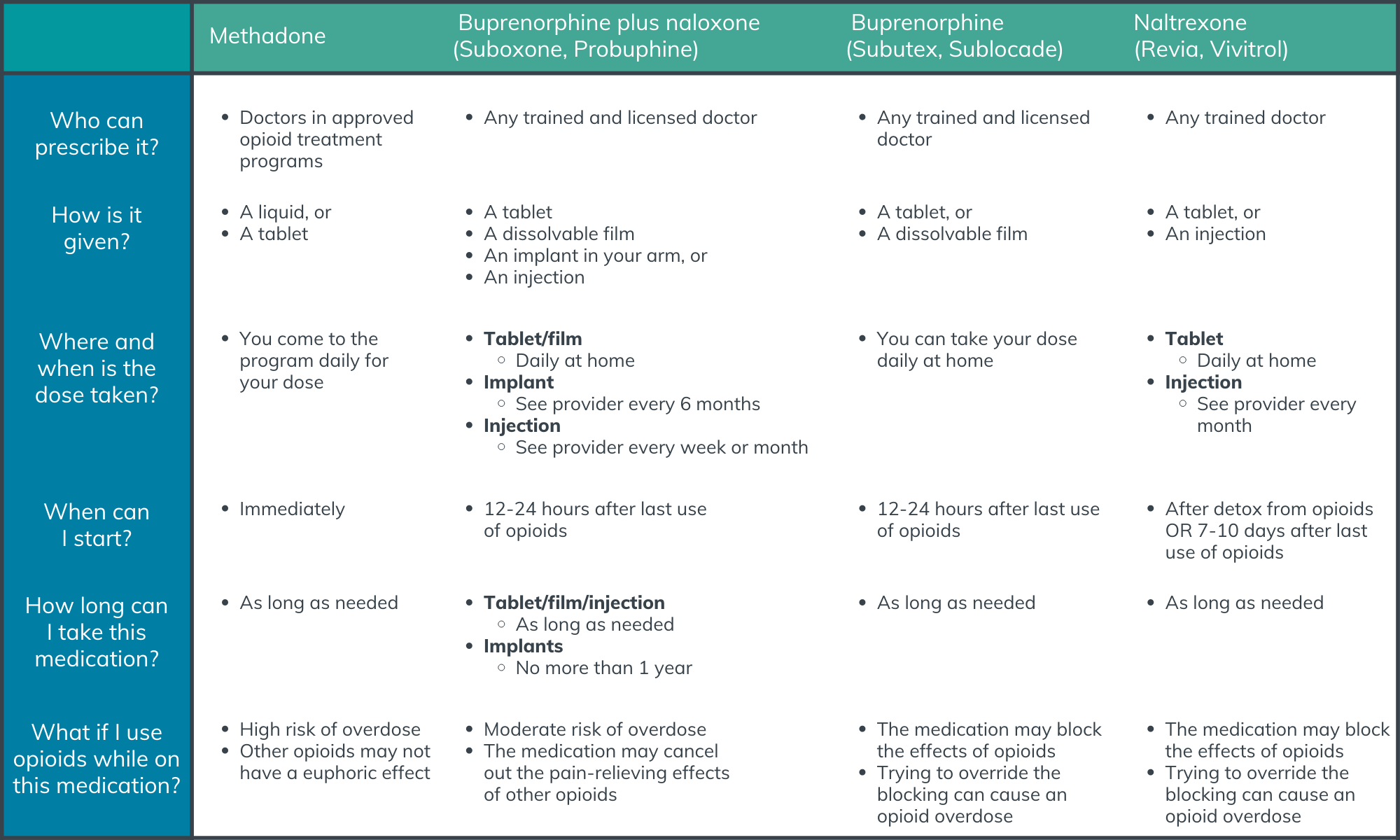
Figure 1.
WHAT DID THIS STUDY FIND?
Study participants with no history of opioid use disorder medications were generally opposed to this treatment modality.
Of the sixteen participants that had no prior experience with opioid use disorder medications, twelve (75%) were explicitly opposed to them whereas only four of the 24 participants (17%) with prior experience with these medications expressed opposition. Those who were opposed to opioid use disorder medications in general cited concerns of financial gain for pharmaceutical companies and replacing one addiction with another.
Preference for short- or long-acting medications differed based on prior opioid use disorder treatment experience.
When given a choice between a long-acting injectable form or a short-acting form of an opioid use disorder medication, 16 (40%) preferred the injectable formulation. When given a choice between a long-acting implant and a short-acting form of an opioid use disorder medication, 12 (30%) preferred the implantable formulation. On the contrary, 50% of participants were not willing to use an injectable formulation and 55% were not willing to use an implantable formulation of opioid use disorder medication. Of those with no experience with opioid use disorder medications and who expressed general opposition, 50% preferred either injectable of implantable medication to short-acting medication, primarily citing decreased misuse potential associated with long-acting formulations. Of those with previous experience on medications for opioid use disorder, 46% expressed preference for injectables and 25% expressed preference for implants over short-acting forms of medication.
Medication-related beliefs were generally impacted by previous experiences.
Participants who had previous experience with short-acting medications preferred this formulation, with many stating they were more comfortable and familiar with this form of the drug. Some people were skeptical about the implant, expressing a concern that the implant could be used as a tracking device or as a means for social control. Others had their own experiences, or experiences of family members or friends, with different types of medications in long-acting formulations (e.g., birth control) which positively or negatively impacted their beliefs about long-acting medications for opioid use disorder.
There were tradeoffs identified that were related to the burden of the medication.
Medication-related burden was primarily represented by convenience, medication characteristics, potential side effects, and structure and support through clinic visits. A majority of participants found long-acting formulations more convenient than the short-acting oral formulations, citing the burden of daily clinic visits interfering with daily routines, travel, vacations, work, and childcare. Long-acting formulations also ensured treatment compliance, as several participants cited difficulty remembering to take their medications on a daily basis. On the contrary, a subset of participants appreciated the accountability, structure, and access to extra services (e.g., individual and group counseling) offered by daily visits to the clinic.
Many participants cited injectables as potential cues (i.e., “triggers”) for cravings to use opioids, especially those who had used opioids intravenously. There was mention that weekly injectables may be more triggering than monthly injections due to more frequent injections. In regard to implantable medications, there was concern over pain and discomfort from the procedure to place and remove the implant. For long-acting forms of opioid use disorder medications in general, concern was raised over how episodes of acute pain would be treated as well as how side effects would be addressed when the medication cannot be removed from the body after administration.
There were pros and cons in relation to medication-taking practices.
Medication-taking practices were largely framed by diversion and misuse potential, patient autonomy, and duration of treatment. Some participants liked the idea of taking a medication that has decreased potential for misuse and diversion, while others viewed long-acting formulations as a loss of control over medication dosage and duration of treatment. Specific to preferring injectable to implantable formulations, some participants stated that the injectable form was a better option because it was a more stable treatment option whereas the implant could be removed from the body. Participants also noted that the six-month duration of the implant would be too permanent compared with weekly or monthly injectables, thus limiting patient autonomy.
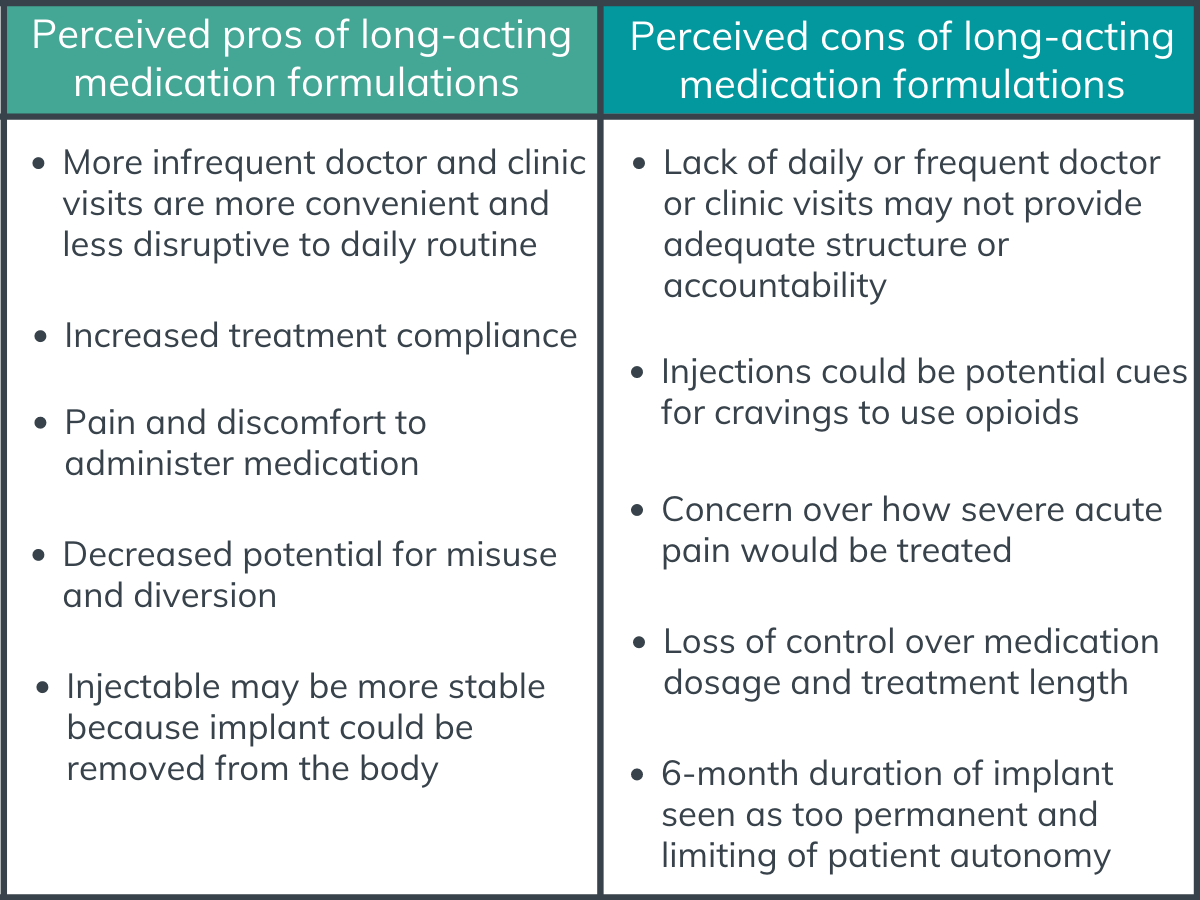
Figure 2. Some perceived pros and cons of long-acting medication formulations according to study participants.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?
Similar to previous studies on preferences for long-acting or short-acting formulations of opioid use disorder medications, preferences and perceptions varied widely based on each participant’s individual experiences, goals, and values. No prior experience with opioid use disorder medications was related to opposition to these medications, although this subset of participants was more open to long-acting formulations due to decreased misuse potential.
Injectable buprenorphine has been shown to reduce opioid use and clinicians are interested in long-acting formulations for their capacity to ensure patients are consistently receiving their medication as well as the potential for a significant reduction in medication misuse or diversion. However, this study illustrates that there are likely a wide range of preferences based on an individual’s needs, experiences, and treatment goals. Access to different formulations of medications should be expanded to match these individual characteristics.
A menu of options for medications is likely to increase utilization among those with opioid use disorder by making the treatment more attractive and acceptable, even among those that are not necessarily seeking treatment. Clinicians can engage in shared decision making with their patients to facilitate this matching of individual characteristics with medication formulation. Integration of peer support into this shared decision-making process could strengthen the trust between patient and provider, further improving treatment outcomes.
Even though this is a cross-sectional study (occurs at one point in time), it is likely that the study participants’ preferences for formulations of opioid use disorder medications are dynamic and will change over time. For instance, a person may prefer the daily structure that is accompanied by a short-acting formulation early in the treatment process but prefer the convenience of a long-acting formulation later in the treatment process. Recovery from opioid use disorder can be a long-term proposition and retention of patients in treatment is critical to improve outcomes. Expanding access to different types and formulations of medications may not only increase utilization among those with opioid use disorder, but it may also improve retention in treatment for individuals whose preferences change over time.
Negative views of opioid use disorder medications in general among many of the study participants highlights the need for practice- and policy-level interventions, including adopting non-stigmatizing language to describe opioid use disorder and medications used to treat this health condition. An interesting finding in this study was that no prior experience with opioid use disorder medications was related to opposition to these medications, although this subset of participants was more open to long-acting formulations due to decreased misuse potential. Expanding access to long-acting formulations may make opioid use disorder medications more acceptable and attractive to those who were previously opposed to this treatment option.
- LIMITATIONS
-
- This qualitative study has a small sample size, as is often the case with qualitative studies. Because the sample is small and participants self-selected into the study, how representative the opinions of this group is of the larger population of people who might benefit from opioid use disorder medications is unknown and should be viewed cautiously. More specific results related to gender, race, geography, or prior opioid use disorder treatment experience should be interpreted also with great caution.
- While authors targeted non-treatment seeking individuals as well as treatment seeking individuals, 90% of the final sample had a history of opioid use disorder treatment. Also, only 25% of study participants reported substance use within the past week, bringing into question the presence of active opioid use disorder among those who were not seeking treatment.
- Some study participants who had access to the internet were emailed an informational tool on the different forms of short- and long-acting medications for opioid use disorder, including naltrexone. Due to naltrexone being an opioid antagonist with a very different pharmacological profile from methadone and buprenorphine, exposure to this information tool could bias perceptions of long-acting forms of buprenorphine.
- The interview guide did not include a question directly assessing general opinions of opioid use disorder medications among study participants, even though this likely served as a significant predictor in preferences between short- and long-acting formulations.
BOTTOM LINE
- For individuals and families seeking recovery: Long-acting formulations of opioid use disorder medications, such as injectables and implants, are FDA-approved treatment options for opioid use disorder. This study shows that preferences and perceptions of these new formulations varied widely based on each participant’s individual experiences, goals, and values. Through shared decision-making, individuals considering medications for opioid use disorder should discuss all options with their provider so that the medication selected provides the best fit based on individual characteristics and stage of stabilization and recovery.
- For treatment professionals and treatment systems: Long-acting formulations of opioid use disorder medications, such as injectables and implants, are FDA-approved treatment options for opioid use disorder. This study shows that preferences and perceptions of these new formulations varied widely based on each participant’s individual experiences, goals, and values. Clinicians are interested in long-acting formulations for their potential to increase patient compliance and decrease medication misuse or diversion. However, clinicians should employ a shared decision-making technique with patients to match the type and form of the medication with a patient’s preferences, needs, and treatment goals. In addition, patients that are generally opposed to opioid use disorder medications may be more open to long-acting formulations.
- For scientists: Long-acting formulations of opioid use disorder medications, such as injectables and implants, are FDA-approved treatment options for opioid use disorder. This qualitative study shows that preferences and perceptions of these new formulations varied widely based on each participant’s individual experiences, goals, and values. Unfortunately, the sample size was too small to better understand how to make these medications more attractive to individuals based on demographics. Studies with larger sample sizes could inform targeting interventions to increase uptake. Also, further qualitative research is needed to explore the experiences and preferences of people who have actually used long-acting formulations of medications for opioid use disorder and longitudinal research can help test the long-term effects of these medications.
- For policy makers: Long-acting formulations of opioid use disorder medications, such as injectables and implants, are FDA-approved treatment options for opioid use disorder. This study shows that preferences and perceptions of these new formulations varied widely based on each participant’s individual experiences, goals, and values. Importantly, no prior experience with medications for opioid use disorder was related to opposition to these types of medications, although this subset of participants was more open to long-acting formulations due to decreased misuse potential. Increasing access to long-acting formulations, perhaps through legal mandates, removing prescription barriers, or expanding insurance coverage, has the potential to increase utilization of these medications and decrease the stigma attached to them.
CITATIONS
Saunders, E. C., Moore, S. K., Walsh, O., Metcalf, S. A., Budney, A. J., Scherer, E., & Marsch, L. A. (2020). Perceptions and preferences for long-acting injectable and implantable medications in comparison to short-acting medications for opioid use disorders. Journal of Substance Abuse Treatment, 111, 54–66. doi: 10.1016/j.jsat.2020.01.009

