For opioid use disorder patients with more challenges, a tailored psychosocial intervention boosts the effects of buprenorphine and methadone
Agonist medications for opioid use disorder, such as buprenorphine (often prescribed in combination with naloxone, commonly known by its brand name Suboxone) and methadone, are effective first-line treatments. When comprehensive support is provided alongside these medications, studies have shown that adding other behavioral treatments does not improve opioid use outcomes. For those with more severe opioid use disorder, however, the benefits of lifesaving medications may not be enough to help individuals get traction in recovery. This paper tested whether adding personalized, comprehensive psychosocial support to medication management improved opioid use disorder outcomes.
WHAT PROBLEM DOES THIS STUDY ADDRESS?
Agonist medications for opioid use disorder, such as buprenorphine (also known by its common brand name Subutex, and when combined with naloxone is also known by the common brand name Suboxone) and methadone are effective first-line treatments, but many individuals continue to use opioids and other drugs after starting these medications. Marsden and colleagues tested whether adding a psychosocial intervention tailored to individual patients that included aspects of cognitive behavioral therapy and contingency management to agonist medication could more effectively help patients achieve abstinence from drugs and improve their quality of life compared to medication alone. Given that such an intervention requires more resources, authors also examined whether adding this psychosocial intervention led to healthcare cost savings over the long-term.
HOW WAS THIS STUDY CONDUCTED?
This was a randomized controlled trial conducted at a community addiction clinic in London, England. Participants were aged 18 years or older, met criteria for opioid or cocaine dependence (based on the Diagnostic and Statistical Manual of Mental Disorders, 4th edition, or DSM-IV) or both, in the past 12 months, and were voluntarily seeking continued oral maintenance opioid agonist therapy, which they had been prescribed for at least 6 weeks. Two-thirds were taking methadone and one-third were taking buprenorphine. All participants were “treatment resistant,” meaning they had used illicit or non-prescribed opioids or cocaine on one or more days in the past 28 days at study screening. Participants were randomized to receive either a personalized psychosocial intervention comprising a flexible toolkit of psychological-change methods, including contingency management to reinforce abstinence, recovery activities, and clinic attendance, in addition to treatment as usual (experimental group), or received treatment as usual only (control group). The primary outcome was treatment response at 18 weeks, which was defined as abstinence from illicit and nonprescribed opioids and cocaine in the past 28 days, as measured with treatment outcome profiles and urine drug screening. The authors employed intent-to-treat analyses, meaning participants who did not complete treatment were treated as “non-responders.”
Psychosocial change was also explored using the following questionnaires administered at baseline, and 18-week follow-up: 1) the Montreal Cognitive Assessment (MoCA), 2) the Patient Health Questionnaire (PHQ-9), which gauges depressive symptoms, 3) the Generalized Anxiety Disorder scale (GAD-7), 4) the Work and Social Adjustment Scale (WSAS), a measure of general, day-to-day functioning, and 5) the EuroQol measure of health-related quality of life (EQ-5D-3L). EQ-5D-3L data were also used to calculate quality-adjusted life-years (QALYs) – a measure of disease burden that includes both quality and quantity of life lived.
The cost-effectiveness of the intervention was also explored in terms of cost savings to the UK National Health Service (NHS) per unit of improvement in treatment outcomes. To do this, the authors calculated incremental cost-effectiveness ratios by dividing the difference in mean costs of providing treatment between the two groups by the difference in mean outcomes. Additional calculations then allowed the authors to explore whether the psychosocial intervention or treatment as usual were the optimal choice at different cost thresholds for each added unit of quality-adjusted life-years.
WHAT DID THIS STUDY FIND?
In terms of treatment response – defined by the authors as no reported use of opioids or cocaine during the 28 days before an 18-week follow-up interview, and one or more negative urine drug tests for heroin and cocaine (and no positive tests) over the same period – 22 (16%) of 135 patients in the psychosocial intervention group (experimental group) had a treatment response, compared with nine (7%) of 135 in the control group. This difference was statistically significant, with an odds ratio of 1.20, meaning participants in the experimental group were 1.2 times more likely to demonstrate a treatment response compared to those in the control group. Participants in the psychosocial intervention group also reported significantly more opioid-abstinent and crack cocaine-abstinent days than those in the control group during the 28 days before the final follow-up interview.
At 18 weeks, mean Work and Social Adjustment Scale scores were significantly lower in the psychosocial intervention group than in the control group, suggesting less impairment in general functioning. However, no between-group differences were observed for cognitive function, depression, or anxiety symptoms.
Although the cost of delivering the psychosocial intervention was greater than the cost of delivering treatment as usual by an average of £561 per participant, the psychosocial intervention was more cost-effective compared to treatment as usual alone. This is because it was able to affect greater treatment response and improvements in quality of life leading to reduced need for costly health care services. Societal costs, including costs of criminal activity and National Health Service and personal social services costs, did not differ significantly between groups.
Also of note, interest in the study was less than expected. The authors struggled to recruit participants and were ultimately not able to recruit their target sample. It is not clear whether this reflected a lack of interest from potential participants in the psychosocial intervention being offered, or more a general lack of interest in participating in research.
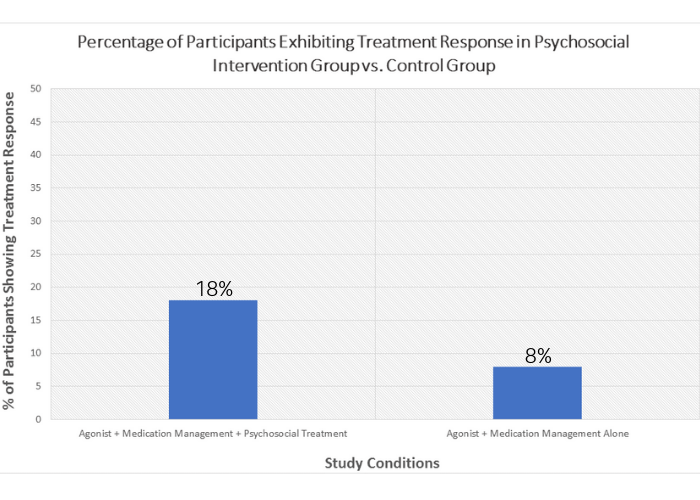
Figure 1. Treatment response rates by group. Participants receiving the psychosocial intervention had greater treatment response (defined as abstinence from illicit and nonprescribed opioids and cocaine in the past 28 days) compared to the control group, which did not receive the psychosocial intervention.
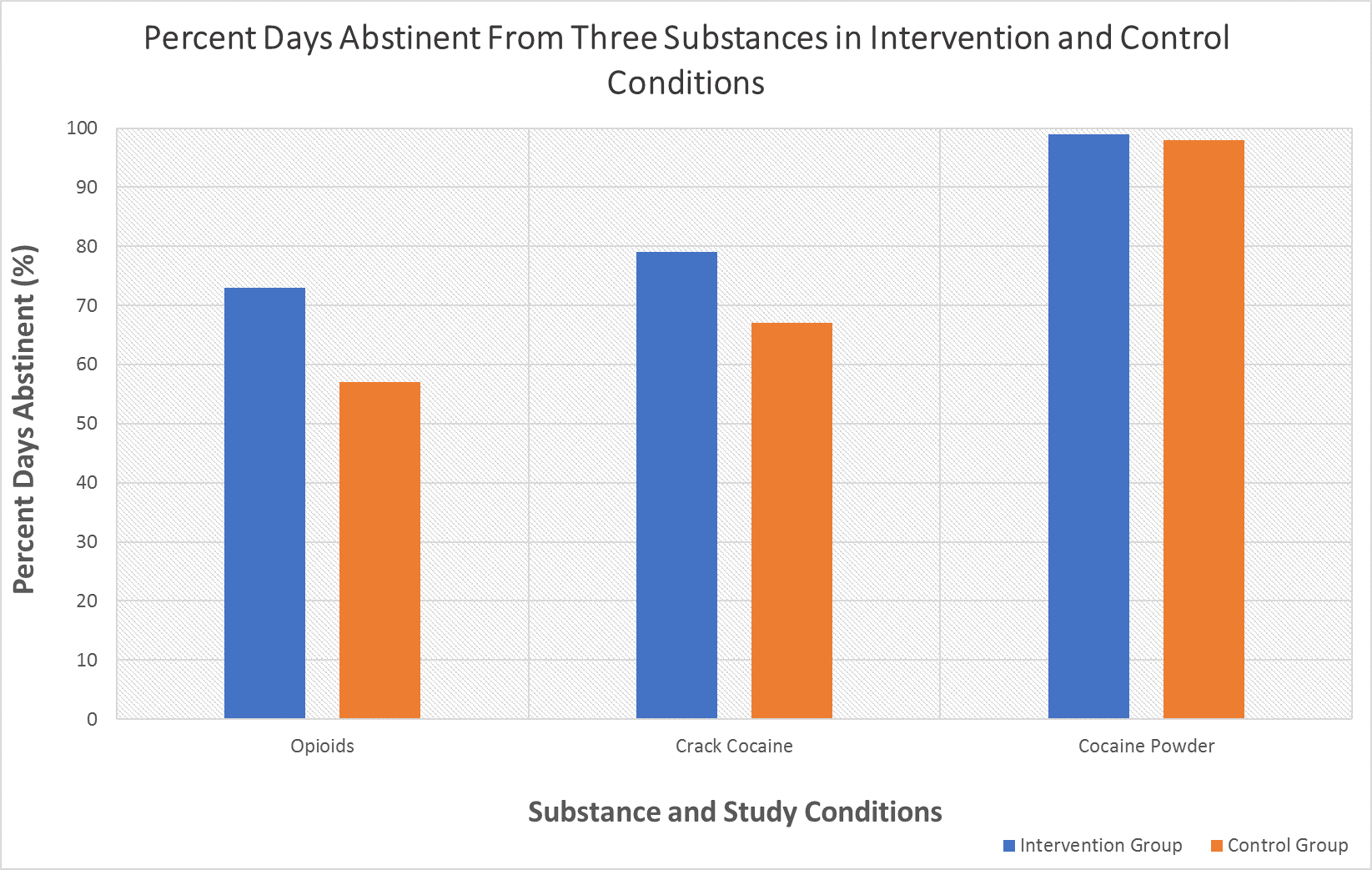
Figure 2. Three substances compared by study condition. Differences in percent days abstinent between the intervention group and control group for each substance. Opioids and crack cocaine demonstrated significantly more percent days abstinent in the intervention group, while cocaine powder did not vary significantly between conditions.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?
While the observed between-group differences were statistically significant, it should be noted that overall the number of participants demonstrating treatment response to the psychosocial intervention was fairly small (i.e., 16%). At the same time, this was a treatment resistant sample (i.e., individuals already taking opioid use disorder medications and continuing to use substances) who were ostensibly dealing with several life challenges (e.g., 90% were unemployed) and coping with a hard-to-treat disorder, so it could be argued this is a meaningful response. Nevertheless, it was still more than double the positive treatment response rate compared to the treatment as usual control group patients.
Based on previous research, the UK National Institute for Health and Care Excellence (NICE) currently recommends that cognitive behavioral therapy should not be offered routinely to those receiving opioid agonist therapy. This recommendation is based on the average treatment effect from samples recruited at admission or during treatment. These authors’ findings, however, suggest that personalized psychosocial interventions may have some therapeutic and cost-saving benefit when administered to individuals not responding well to medication treatment. A personalized approach in which specific behavior-change interventions are tailored to individuals’ needs could prove to be a more fruitful approach than untailored cognitive behavioral therapy alone.
It is noteworthy that the participants treated in this study had high substance use disorder severity and appeared to benefit from extra psychosocial support. This finding highlights the need to look for individual factors that may influence treatment response. Ling and colleagues, for instance, found that the addition of cognitive behavioral therapy and contingency management to opioid agonist therapy did not produce significantly greater reductions in heroin use. This research, however, highlighted the fact that medication management involved numerous visits with clinical staff and providers. It is probable that these interactions made patients feel supported and cared about, and ensured they received addiction recovery suggestions, referrals, and advice. In short, medication management may have been providing patients with the kind of support offered by cognitive behavioral therapy, and thus suggests that those not receiving intensive medication management (e.g., a patient getting prescription refills with little facetime with providers) may benefit markedly from cognitive behavioral therapy in addition to medication. This idea is also supported by the work of Carroll and Weis.
- LIMITATIONS
-
- To the authors’ credit, they made every effort to complete 18-week follow-ups with participants, even if that meant this follow-up did not occur until up to week 24. This, however, meant that the estimate of the psychosocial intervention’s efficacy was conservative because there was a tendency for those with long follow-up times to be identified when they re-presented for further opioid agonist therapy after relapse.
BOTTOM LINE
- For individuals and families seeking recovery: These findings suggest that individuals with more severe substance use disorder could benefit from psychosocial interventions in addition to medications like Suboxone. If taking medications alone, try to find treatment that offers check-ins and monitoring, rather than just brief provider visits to refill prescriptions.
- For treatment professionals and treatment systems: The authors’ findings suggest that those with more severe substance use disorder stand to benefit from tailored and specific psychosocial interventions when it is offered in addition to pharmacotherapy. If providing pharmacotherapy alone, patients will likely benefit from medication management treatment that includes frequent check-ins, discussions about progress, and patient monitoring, rather than merely brief prescriber visits for prescription refills.
- For scientists: The authors’ findings suggest that those with more severe substance use disorder can benefit from specific and tailored psychosocial interventions when they are offered in addition to pharmacotherapy. Work to-date suggests individual factors and the intensity of medication management influence treatment response to pharmacotherapy with and without cognitive-behavioral interventions for substance use disorder. Research is critically needed exploring possible moderators of treatment response to pharmacotherapeutic approaches as stand-alone interventions, and when combined with cognitive-behavioral interventions.
- For policy makers: These findings suggest that individuals with more severe substance use disorder could benefit from tailored and specific psychosocial interventions in addition to medication. Creating access to life-saving medications like Suboxone as well as psychosocial support through psychotherapy has the potential to dramatically improve public health.
CITATIONS
Marsden, J., Stillwell, G., James, K., Shearer, J., Byford, S., . . . Mitcheson, L. (2019). Efficacy and cost-effectiveness of an adjunctive personalised psychosocial intervention in treatment-resistant maintenance opioid agonist therapy: A pragmatic, open-label, randomised controlled trial. Lancet Psychiatry, 6(5), 391-402. doi: 10.1016/s2215-0366(19)30097-5

