Extended release versions of buprenorphine: Patient perspectives on pros and cons
Options for opioid agonist therapy are expanding with the development of injectable, extended-release 1-week, 1-month, and 6-month formulations of buprenorphine (also known by the common brand names Subutex or Suboxone when combined with naloxone). There is an assumption that patients will welcome these new formulations, but what do patients themselves think?
WHAT PROBLEM DOES THIS STUDY ADDRESS?
There is strong evidence that opioid agonist medications help reduce opioid use and overdose risk, but there have been challenges with uptake of life saving medications like buprenorphine. These challenges include lack of prescribers licensed to prescribe this medication, risk of missing doses and going into opioid withdrawal, and risk for medication diversion and misuse. As such, there is much interest among clinicians about extended-release buprenorphine formulations lasting 1-week, 1-month, and 6-month that are now available in many countries. At the same time, the people who would take these medications (i.e., people with opioid use disorder) are seldom consulted.
In a prior study, Neale and colleagues found that a sample of individuals who had lived experience taking opioid agonist medication identified both benefits and concerns associated with depot injection and implant formulations of buprenorphine. Benefits included, dose stability, their potential for helping individuals break rituals and habits associated with drug use, feeling normal, and being able to get on with everyday life. Concerns included difficulty stopping treatment and reduced choice and control once the injection or implant has been administered, and less social interaction. In this study, they sought to build on their previous work attempting to understand which formulations would be preferred and why.
HOW WAS THIS STUDY CONDUCTED?
This was a qualitative study with 36 participants (26 men and 10 women, aged 24-63 years) who were taking daily oral opioid agonist medications for opioid use disorder (e.g., methadone or buprenorphine) or taking heroin daily without opioid agonist medications. All were asked for their views on weekly, monthly, and six-monthly formula durations of buprenorphine. Responses were audio-recorded, transcribed, and analyzed.
This research was conducted in London, UK, during 2018 before any prolonged-release opioid agonist therapy formulations were approved in Europe. As such, participants’ opinions were purely hypothetical rather than based on personal experience. 12 participants were taking prescribed oral methadone, 12 were taking prescribed oral buprenorphine, and 12 were using heroin daily but not currently prescribed any opioid agonist medication. Just over a third were collecting opioid agonist medication from a pharmacy daily, five were collecting opioid agonist medication 2-3 times a week, and three were collecting opioid agonist medication weekly. Over half reported that they were currently taking heroin.
The researchers let participants know that the study was being funded by a pharmaceutical company developing a prolonged-release buprenorphine formula. With the help of a written guide, the interviewer gathered information about participants’ personal circumstances, substance use, treatment history, and views on prolonged-release buprenorphine. To facilitate discussion, participants were provided with a basic overview of the features of prolonged-release buprenorphine depot injections. Participants also had the opportunity to inspect a prototype of the depot injection prefilled syringe if they wanted.
Next, participants views on medication duration were solicited, with an invitation. To this end, to comment on why people might or might not want to receive a 1-week, 1-month, or hypothetical 6-month buprenorphine depot medication, which duration they would personally prefer, and why people might want to change between medication durations. The researchers discussed a hypothetical 6-month depot medication rather than what is in reality a 6-month implant so as to maintain a focus on duration of medication effects.
WHAT DID THIS STUDY FIND?
Most participants liked the idea of longer duration medicines.
In line with the authors’ previous work on this topic, most participants felt that having medications of different durations was positive since this gave them choice and meant that treatment could be better tailored to meet their needs.
Participants consistently reported that a depot injection that lasted a month, and ideally 6 months, would be more valuable than a weekly depot injection in terms of increasing their ability to live a ‘normal life.’ Many participants also explained how the longer (1-month or 6-month) formulations would remove the embarrassment they experience having to take their medication in a pharmacy, and alleviate the stress of getting to so many doctor’s appointments.
A number of participants also noted these formulations would provide them with welcome respite from the physical symptoms of withdrawal (being ‘sick’) and the constant psychological worry that they might become ‘sick’. In turn, they said that this would have an important further benefit of reducing the time that they spent thinking about obtaining and using drugs. This general sentiment was frequently expressed in the previous study by these authors, which found that the dose stability offered by longer buprenorphine formulations was an important benefit.
Some specific perceived benefits of the 1-week formulation were noted.
Participants repeatedly noted that the weekly depot injection would be best for initiating treatment, allowing people to ‘try the medication out’ before committing to it. They also pointed out that having a shorter initial treatment period would give providers the opportunity to confirm that the patient was committed to treatment, while also giving patients the opportunity to see how they respond to the dose. Participants also noted that the 1-week depot injection would be better for people who were apprehensive about taking an opioid agonist medication. Participants also felt that the weekly depot injection had the advantage of facilitating more regular contact with healthcare providers.
Potential applications were suggested for people in prison or at risk for going to prison.
Several participants suggested that longer formulations would be beneficial in prisons. For example, they said that these formulations would reduce illicit substance use, medication diversion, and prison violence, and this would ultimately save the prison service resources. In addition, one participant reported that a 6-month depot injection could have a useful role as an ‘enforced’ community treatment—providing a better alternative to a prison sentence for people who broke the law.
Some important concerns were also raised.
Some participants stated that the weekly formulation would not give people time to make any fundamental life changes before the medication ended, so making it relatively easy for people to go back to using street drugs. Other participants argued that nobody should be maintained on opioid agonist medication for 6 months as the goal must always be reduction leading to abstinence, while some voiced concerns that a 6-month formulation might release too much medication at once, causing people to overdose, or have other long-term harmful side effects. Concerns were also raised that longer formulations would lead to less contact with treatment providers, and thereby diminish recovery support.
One concern raised in the authors’ previous work on this topic, but not highlighted in this study, were participant worries about medical complications associated with receiving a depot injection. Participants in the previous study (which explicitly sought opinions about more invasive implant buprenorphine delivery systems as well as depot injections) were particularly concerned about potential medical complications arising from implants. They also expressed concerns and mistrust about having “a little bot,” “nanite,” or “microchip” implanted inside them. It is possible participants in the study reviewed here would have also expressed such concerns were they explicitly asked about implants.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?

Figure 1.
Previous empirical research strongly supports the capacity of depot injection buprenorphine to reduce opioid use, and importantly, that adverse buprenorphine injection events are relatively rare. These injectable buprenorphine formulations are appreciated by clinicians for a number of reasons, such as that they ensure patients are consistently receiving their medication, and they dramatically reduce the risk of medication diversion or misuse.
The participants in this and a previous qualitative study by the study authors also appreciated these attributes, while further highlighting a number of other important potential benefits of long-acting forms of buprenorphine. These included better control over treatment planning, avoiding stigma and embarrassment routinely experienced as a result of having to take medications in a community pharmacy in front of other customers, more free time and increased ability to lead ‘a normal life,’ and avoiding withdrawal symptoms and places where there is a chance of running into drug dealers (e.g., at methadone clinics).
At the same time, important concerns about these formulations were identified by the participants, particularly with regard to the 6-month formulation. Concerns were expressed that this formulation reduced treatment flexibility (i.e., not being easily able to stop the medication if you change your mind). Concerns were also raised about risk of long-term, harmful side effects, though it should be noted that in reality, 6-month formulations actually utilize implants that can be easily removed by providers in the event of an adverse reaction. Participants were also concerned that longer formulations (1-month and 6-month) might not last as long as the drug developers promised, and also would reduce their contact with healthcare providers.
On the whole, the authors found that their study participants appreciated the utility of the different formulations of long-acting buprenorphine formulations. Ultimately, having these new treatment options is a good thing, however, it will be important that healthcare providers consult with individual patients about their needs and preferences so they can receive the formulation that best suits them, and maximizes their chances of recovery. Also, different formulations may be indicated at different stages of recovery, and as suggested by a number of the study participants, it may be that some patients may benefit from starting with shorter duration depot injections (1-week or 1-month) before transitioning to 6-month implants.
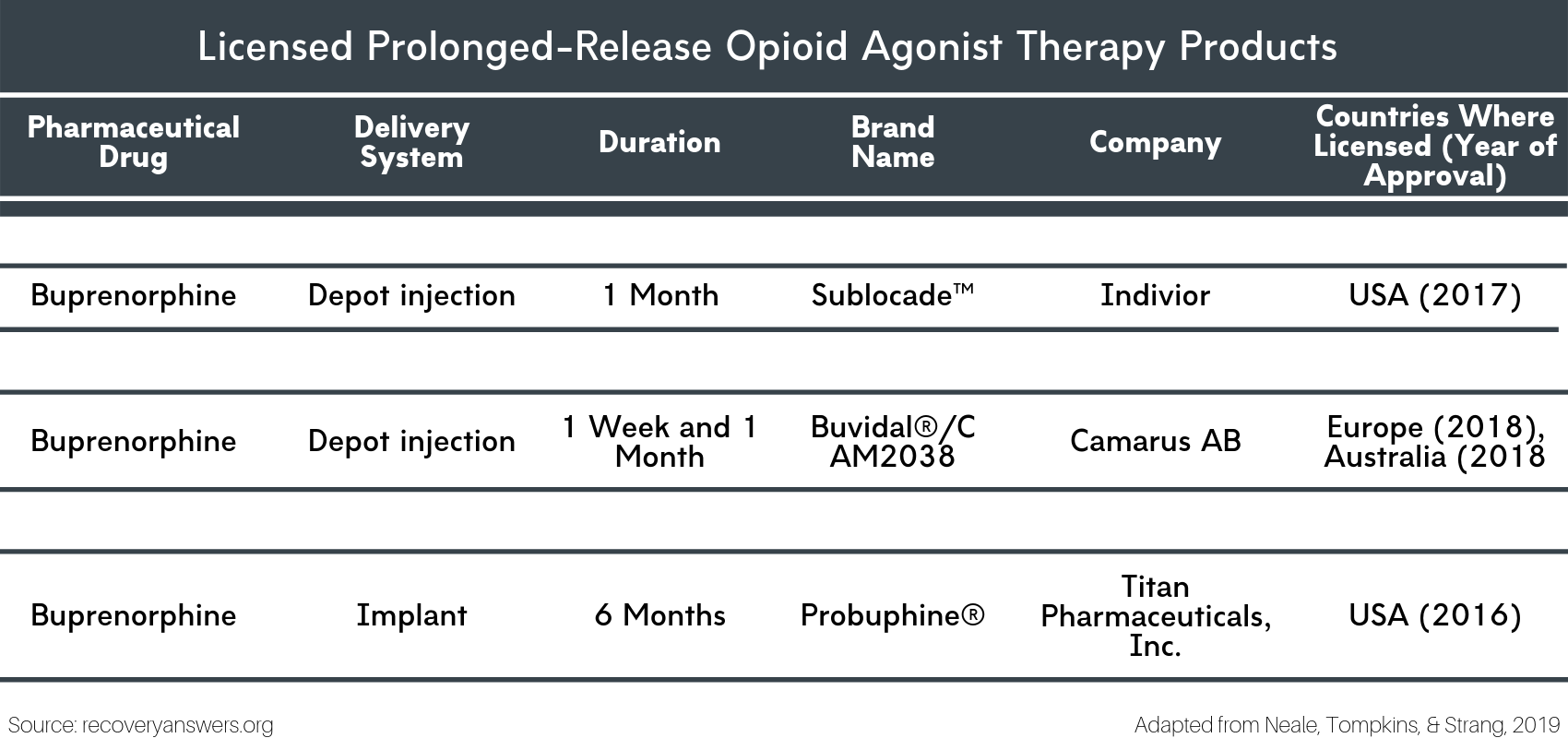
Figure 2.
- LIMITATIONS
-
- As the authors note, participants were asked to discuss forms of prolonged-release opioid agonist medications not available in the UK at the time of the interviews. What participants think about and expect from a treatment they have never actually tried may be very different from what they might report after they have personally experienced it.
- As the authors note, this study was conducted at a single site in London. It is possible that peoples’ opinions about these formulations differ by geographical location (e.g., city, state, country). Consequently, we must be careful about generalizing these findings to other locations.
- Also, the sample size was too small to reliably identify any complex sub-group differences based on variables such as gender, race, or prior opioid agonist medication experiences.
BOTTOM LINE
- For individuals and families seeking recovery: Opioid agonist medications like buprenorphine are effective for the treatment of opioid use disorder, and 1-week, 1-month, and 6-month buprenorphine formulations are now available in many countries. This study showed that, on the whole, people who would use these medications largely have positive perceptions of them and appreciate their potential to support recovery and improve quality of life. These formulations are well worth considering given the potential benefits they offer. Talk to your prescriber and/or your insurance company about these medication options.
- For treatment professionals and treatment systems: Opioid agonist medications like buprenorphine are effective for the treatment of opioid use disorder, and 1-week, 1-month, and 6-month buprenorphine formulations are now available in many countries. This study showed that, on the whole, people who would use these medications largely have positive perceptions of them and appreciate their potential to support recovery and improve quality of life. These new formulations have the potential to better support patients’ recovery efforts and improve their quality of life, and should be discussed with patients with opioid use disorder.
- For scientists: Opioid agonist medications like buprenorphine are effective for the treatment of opioid use disorder, and 1-week, 1-month, and 6-month buprenorphine formulations are now available in many countries. This study showed that, on the whole, people who would use these medications largely have positive perceptions of them and appreciate their potential to support recovery and improve quality of life. Further qualitative research is needed to explore the experiences of people who have actually used prolonged-release opioid agonist medications. Additionally, although the efficacy of the formulations has been shown in empirical research, more work is needed on the long-term effects of these medications.
- For policy makers: Opioid agonist medications like buprenorphine are effective for the treatment of opioid use disorder, and 1-week, 1-month, and 6-month buprenorphine formulations are now available in many countries. This study showed that, on the whole, people who would use these medications largely have positive perceptions of them and appreciate their potential to support recovery and improve quality of life. It is critical that individuals have access to these life-saving medications, and toward this end, access to these medications can be improved by reducing prescription barriers and requiring insurance companies to cover opioid agonist medications.
CITATIONS
Neale, J., Tompkins, C. N. E., & Strang, J. (2019). Prolonged-release opioid agonist therapy: qualitative study exploring patients’ views of 1-week, 1-month, and 6-month buprenorphine formulations. Harm Reduction Journal, 16(1), 25. doi:10.1186/s12954-019-0296-4

