Recent federal policy changes associated with expansion of buprenorphine prescribing with important rural-urban differences
Most people with opioid use disorder do not receive treatment and even fewer receive evidence-based treatment with medications. New policies have been enacted to expand access to medication treatment for opioid use disorder. This study used administrative databases in California to measure the impact of 2 federal policies that increased the patient limit for physicians who are appropriately trained and permitted (“waivered”) to prescribe buprenorphine and allowed new types of healthcare professionals to become waivered to prescribe buprenorphine.
WHAT PROBLEM DOES THIS STUDY ADDRESS?
The opioid addiction and overdose crisis in the United States continues to escalate, as more than 105,000 people are estimated to have died from a drug overdose in a 12-month period ending in October 2021, an all-time record with the vast majority of these deaths involving an opioid. Medications for opioid use disorder, such as methadone or buprenorphine (commonly prescribed in formulation with naloxone and known by the brand name Suboxone), have been shown to substantially decrease opioid-related mortality. Despite this evidence, it is estimated that only 11% of individuals who met criteria for opioid use disorder in 2020 received these medications. Reasons for underutilization are complex, but one partial explanation is a gap between treatment need and the ability of clinical providers and systems to provide it (treatment capacity).
There have been several federal policy changes in recent years aimed at increasing treatment capacity for opioid use disorder. These new policies have predominantly targeted buprenorphine expansion, which is a treatment option that can be prescribed similar to other types of medications for other diseases, by an appropriately trained healthcare professional in an outpatient setting and dispensed from a pharmacy. This is in contrast to methadone treatment which can only be delivered in federally licensed and highly structured opioid treatment programs. For qualified healthcare professionals to be able to prescribe buprenorphine, they had to obtain a waiver by receiving specialized training, although these requirements have recently been changed.
Previous studies have shown that recent federal policy changes aimed at expanding buprenorphine access were associated with an increase in the number of waivered providers and the number of buprenorphine prescriptions. In this study, researchers used several administrative databases in California to measure the impact of 2 federal policy changes, specifically examining the uptake of new waivers and their association with buprenorphine prescribing and opioid-related harm at the county level with a focus on rural-urban differences. Findings from this study can shed light on the public health effects of these policy changes beyond changes in buprenorphine prescribing as well as lead to a better understanding of which types of waivers are most important in rural and urban buprenorphine expansion.
HOW WAS THIS STUDY CONDUCTED?
This study used several administrative databases in California to examine the uptake of new waivers to prescribe buprenorphine created after the implementation of 2 federal policy changes and their association with buprenorphine prescribing and opioid-related harm. The researchers leveraged variability in waiver uptake at the county-level to assess these outcomes with a particular focus on differences between rural and urban counties.
This study measured the impact of 2 federal policy changes that occurred around the same time in 2016 that broadly increased permissions with regard to who could prescribe buprenorphine and how many patients prescribers were permitted to serve at any one time. The first policy change, a final rule from the Substance Abuse and Mental Health Administration, increased the patient limit of physicians already waivered to prescribe buprenorphine from a maximum of 100 to a maximum of 275. The second policy change, part of a Congressional law called the Comprehensive Addiction and Recovery Act, allowed nurse practitioners and physician assistants to become waivered and prescribe buprenorphine for patients with opioid use disorder with a 30-patient limit.
There were 2 primary variables of interest 1. the buprenorphine prescribing capacity rate for new waivers from nurse practitioners and physician assistants and 2. the buprenorphine prescribing capacity rate for new waivers that increased the patient limit for physicians to 275. Notably, a capacity rate is the maximum number of patients that healthcare professionals could treat with buprenorphine, not the actual number of patients being treated. Waiver information, including type, month, and county, was collected from the Substance Abuse and Mental Health Administration (SAMHSA). The researchers wanted to see how different buprenorphine prescribing capacity rates for these waivers at the county-level were associated with outcomes that measured actual buprenorphine prescribing and opioid-related harm.
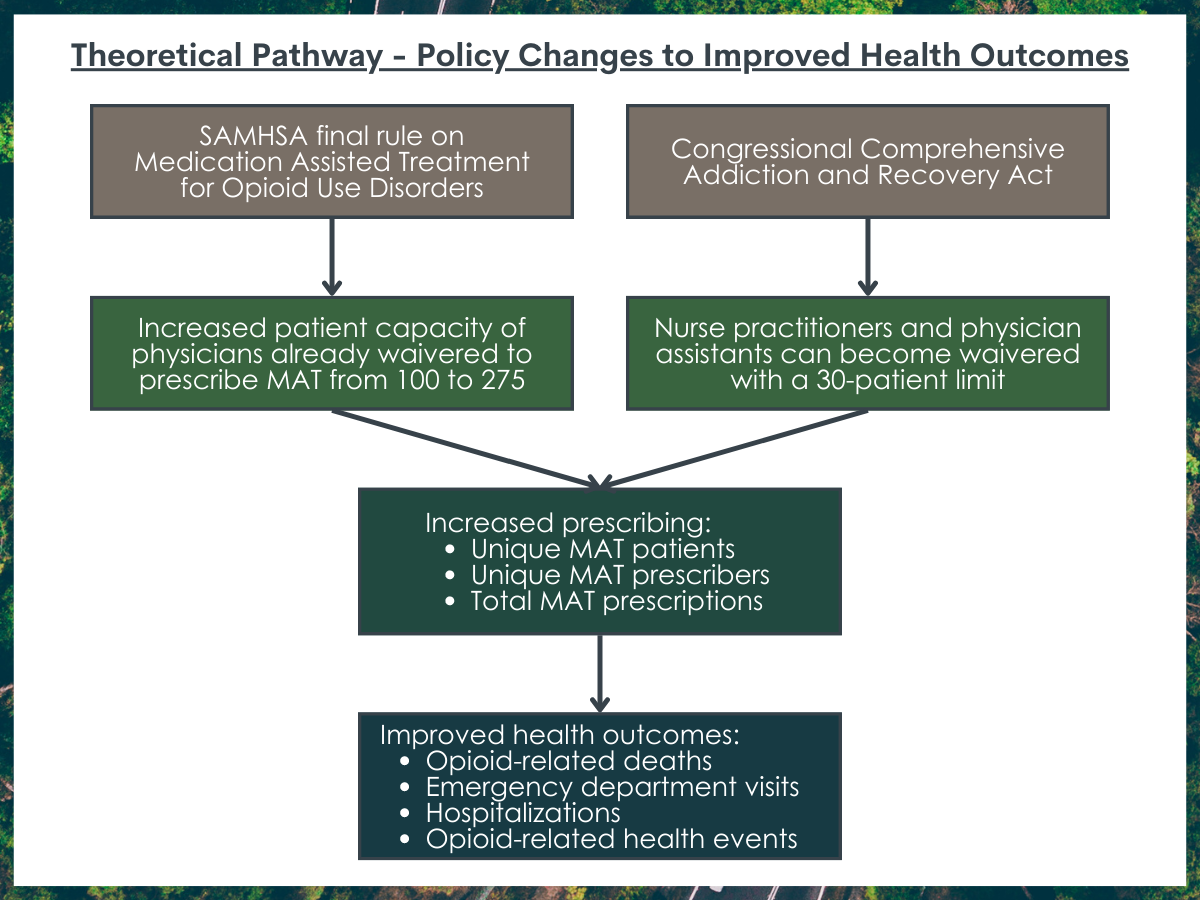
There were 7 outcomes assessed. Three outcomes measured buprenorphine prescribing and included rates of unique buprenorphine patients, unique buprenorphine prescribers, and total buprenorphine prescriptions. These data came from California’s prescription drug monitoring program, called the Controlled Substance Utilization and Evaluation System. Four outcomes measured opioid-related harm and included opioid-related deaths, emergency department visits, and hospitalizations as well as a measure for total opioid-related health events. Mortality data came from the California Department of Public Health and healthcare utilization data came from the California Office of Statewide Health Planning and Development.
The researchers first assessed trends in different types of waivers in California from 2010 to 2018. Then, they used 14 separate statistical analysis models to examine the relationship between the 2 clinical prescribing policy changes and the 7 outcomes of interest. Data were analyzed at the county level and aggregated by quarter.
Since there were many other factors that could affect both the policy prescribing changes and the outcomes, the researchers used several “best-practice” statistical techniques in an effort to isolate the relationship between these 2 new types of waivers and the outcomes measuring buprenorphine prescribing and opioid-related harm.
The researchers also examined the differences between urban and rural counties where metropolitan status was derived from the United States Office of Management and Budget. Several additional analyses were performed to ensure that the pattern of findings resulting from the statistical procedures were consistent and reliable.
WHAT DID THIS STUDY FIND?
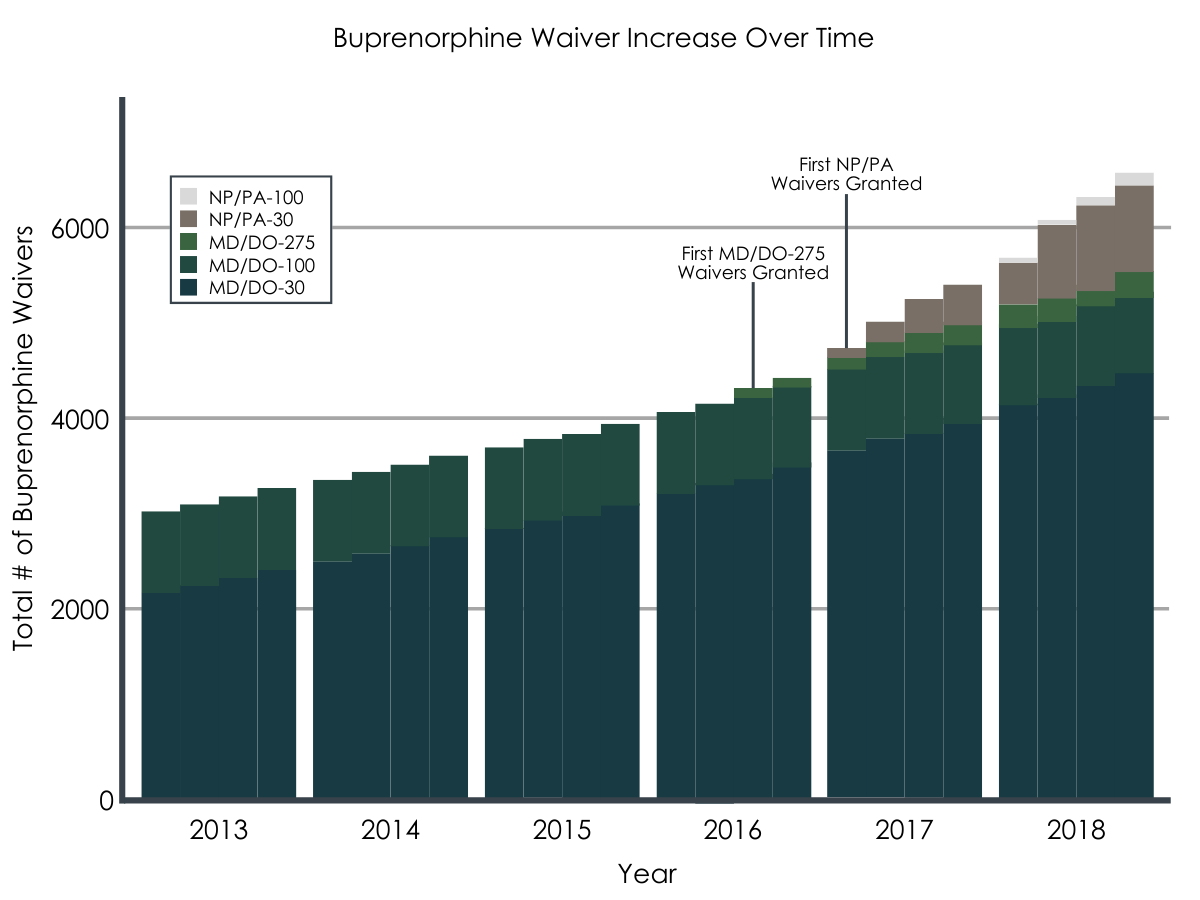
The buprenorphine prescribing capacity increased substantially after the introduction of the new federal policy changes.
The number of waivered providers, and thus the buprenorphine prescribing capacity rate, increased over the study period from 2010 to 2018, with more rapid increases following the policy change that increased the patient limit for some physicians from 100 to 275 (implemented in the 1st quarter of 2016) and the policy change that allowed nurse practitioners and physician assistants to become waivered (implemented in the 3rd quarter of 2016).
The new types of waivers were associated with increased buprenorphine prescribing.
At the county level, each additional new waiver of a nurse practitioner or physician assistant was associated with a 2.2% increase in the buprenorphine prescriber rate, a 1.4% increase in the buprenorphine patient rate, and a 1.9% increase in the total buprenorphine prescription rate. Each additional new waiver that increased a physician’s patient limit to 275 was associated with a 3.1% increase in the buprenorphine prescriber rate, a 4.1% increase in the rate of unique buprenorphine patients, and a 3.1% increase in the total prescription rate.
There were rural-urban differences in buprenorphine prescribing for each new type of waiver.
In rural counties, each additional new waiver of a nurse practitioner or physician assistant was associated with increases in all 3 measures of buprenorphine prescribing, but this new type of waiver was not associated with changes in buprenorphine prescribing in urban counties. Each additional new waiver that increased a physician’s patient limit to 275 was associated with an increase in all 3 measures of buprenorphine prescribing in rural counties and an increase in both the buprenorphine patient rate and the total prescription rate in urban counties.
The new types of waivers were not associated with changes in opioid-related harms.
There were no changes in opioid-related deaths, emergency department visits, and hospitalizations as well as a measure for total opioid-related health events associated with the new types of waivers.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?
Study findings suggest that buprenorphine prescribing capacity increased substantially after the new federal policy changes, and the increased capacity resulting from the new types of waivers was associated with increases in buprenorphine prescribing, with this increase driven by both types of waivers in rural counties and driven by the waiver increasing the patient limit to 275 for urban counties.
Findings suggest that the new waiver that allows nurse practitioners and physician assistants to prescribe buprenorphine will be most impactful on increasing buprenorphine prescribing in rural areas, possibly by increasing access to more prescribers. This is similar to previous research that has suggested that healthcare professionals other than physicians are likely to play an important role in expanding buprenorphine access in rural areas. Specifically, empirical research shows that new waiver uptake in rural areas is driven by nurse practitioners and physician assistants although scope of practice regulations at the state level can facilitate or hinder uptake. This is an important finding because, although access has improved in recent years, small rural communities still experience buprenorphine treatment disparities, possibly due to a lack of physician prescribers. Notably, findings suggest that the new waiver increasing the patient limit to 275 will play an important role in improving buprenorphine access in both rural and urban areas.
An interesting finding in this study is that the new waivers had no impact on opioid-related harms. However, there is strong evidence that increasing buprenorphine treatment among people with opioid use disorder reduces opioid-related mortality and improves other outcomes. Translation of these individual-level benefits to the population level depends on a variety of factors. It is possible that a longer time frame was needed to realize an impact on opioid-related harm, as the new federal policy changes occurred in the early and latter parts of 2016 and the study period ended in 2018. It could also mean that at least some of those individuals who are most likely to experience overdose may not be willing to seek buprenorphine treatment regardless of how available or accessible it may be. Another possibility is that the study findings are subject to the ecological fallacy, where measured impacts may be different when using aggregated data at the county level and individual-level data.
Despite findings that these new federal policy changes were associated with increased waiver uptake among healthcare professionals and increased buprenorphine prescribing, policies addressing the number of waivered providers is just part of the solution. Other research has highlighted myriad barriers to expanding access to buprenorphine. Only half of waivered providers actually prescribe buprenorphine and the majority of waivered providers do not prescribe at their maximum limit. In addition, stigma among healthcare professionals is pervasive and similar to the general population, and many healthcare professionals are not interested or feel ill-prepared in treating opioid use disorder. New buprenorphine practice guidelines were released by the Department of Health and Human Services in April 2021 that no longer require additional training to obtain the waiver. In addition to directly reducing barriers to obtaining the waiver, there is hope that no longer requiring specialized training will send the message to providers that they can appropriately manage opioid use disorder by prescribing buprenorphine and following these patients clinically over time.
- LIMITATIONS
-
- Although the researchers used many statistical techniques to try to isolate and infer a causal connection between the federal policy changes and the outcomes of interest, an actual causal connection, while plausible, cannot be determined. For example, there are likely many initiatives to increase buprenorphine treatment among people with opioid use disorders at the federal, state, and local level that could not be controlled for in these analyses.
- This study uses administrative data which is not collected for research purposes and can sometimes be subject to inaccuracies.
- This study is subject to the ecological fallacy where it cannot be confirmed that the new types of waivers directly contributed to individual-level outcomes, such as the number of unique buprenorphine patients.
- This study was done in California so generalizing these findings to other states or nationally should be done with caution.
- There were limitations in the database used to identify waivered providers, such as whether the provider practiced in more than one county, had retired, or moved to another county during the study period.
BOTTOM LINE
This study used administrative databases from California to examine how new types of buprenorphine waivers introduced through federal policy changes were associated with changes in buprenorphine prescribing and opioid-related harm at the county level. Findings show that buprenorphine prescribing capacity increased substantially after these policy changes. This increased capacity was associated with increases in buprenorphine prescribing, with this increase driven by both types of waivers in rural counties (i.e., by expanding who could prescribe and by increasing the number of patients physician providers could serve at any given time), and driven by the waiver that increased the physician patient limits to 275 for urban counties.
- For individuals and families seeking recovery: Buprenorphine is an effective medication to treat opioid use disorder. Many patients will need support beyond medication treatment, such as counseling and services to address social determinants of health like employment, education, and housing. Buprenorphine treatment can be delivered in a private doctor’s office and the medication can be dispensed by a pharmacy. Due to its promise in addressing the opioid crisis, recent and future federal and state policy changes are likely to target improving access to buprenorphine treatment.
- For treatment professionals and treatment systems: Expanding access to buprenorphine is an important tool in addressing the opioid crisis, and federal policy changes that allowed nurse practitioners and physician assistants to become waivered to prescribe buprenorphine and increased the patient limit for some physicians were associated with increases in buprenorphine prescribing in California. The role of nurse practitioners and physician assistants in expanding access to buprenorphine in rural areas may be especially important to increase access. Even though these policy changes are a step in the right direction, there are still many other barriers to overcome in expanding access to buprenorphine, such as addressing stigma among healthcare professionals and increasing awareness and knowledge around the scientific evidence for medication treatment for opioid use disorder.
- For scientists: This study employs a rigorous analytic approach and found that the new types of waivers were associated with increased buprenorphine prescribing, with important rural-urban differences, but there was no association with opioid-related harm. Future studies should explore why there was no change at the population level when there is strong evidence that buprenorphine treatment reduces opioid-related mortality and improves other outcomes at the individual level. In addition, although these policy changes are a step in the right direction, there are still many other barriers to overcome in expanding access to, as well as utilization of, buprenorphine, and it is uncertain which policies and initiatives are most critical in increasing buprenorphine prescribing and patient uptake and translating these to reducing ultimate opioid-related harm.
- For policy makers: This and other studies have shown that increasing the number of waivered providers is a way to increase buprenorphine treatment among people with opioid use disorder. However, there are myriad barriers to address in addition to increasing uptake of the waiver. A recent federal policy change removed the training requirement to become waivered, but prescribers, including physicians, nurse practitioners, and physician assistants, must still take steps to obtain a waiver and could only treat up to 30 patients without additional training. In addition, there may be statutes and regulations at the state level that restrict this new federal policy change. Non-physician healthcare professionals, such as nurse practitioners and physician assistants, may play an especially important role in improving buprenorphine access in rural areas.
CITATIONS
Rowe, C.L., Ahern, J., Hubbard, A., Coffin, P.O. (2022). Evaluating buprenorphine prescribing and opioid-related health outcomes following the expansion the buprenorphine waiver program. Journal of Substance Abuse Treatment, 132, 108452. doi: 10.1016/j.jsat.2021.108452

