Naltrexone-facilitated Buprenorphine (Suboxone) Discontinuation for Patients in Recovery from Opioid Use Disorder
Buprenorphine is a partial opioid agonist commonly prescribed to individuals with opioid use disorder who are seeking recovery.
While buprenorphine (Suboxone) allows individuals to gradually recover from opioid addiction by diminishing the effects of withdrawal, it still has the ability to cause physical dependence.
When discontinuing buprenorphine, it is hypothesized that patients taking even low doses experience withdrawal discomfort due to loss of k-opioid receptor antagonism (i.e., blocking of the k-opioid receptor), one of the effects of buprenorphine (mu opioid receptor agonism is the other).
Since naltrexone, a medication prescribed for the treatment of alcohol and opioid use disorders, is an antagonist of these receptors, researchers believe that it may help facilitate buprenorphine discontinuation by diminishing withdrawal symptoms.
Dakwar and Kleber conducted an open-label trial to investigate feasibility and tolerability of using naltrexone to aid buprenorphine discontinuation in patients who achieved sustained full remission from opioid use disorder (i.e., patients were prescribed suboxone and were abstinent from illicit or other prescription opioids for at least 1 year) and tapered to 2 mg or less of buprenorphine but were unable to stop altogether. Other eligibility criteria included medical and psychiatric stability, no history of overdose, no other current substance use disorders, and no history of chronic pain.
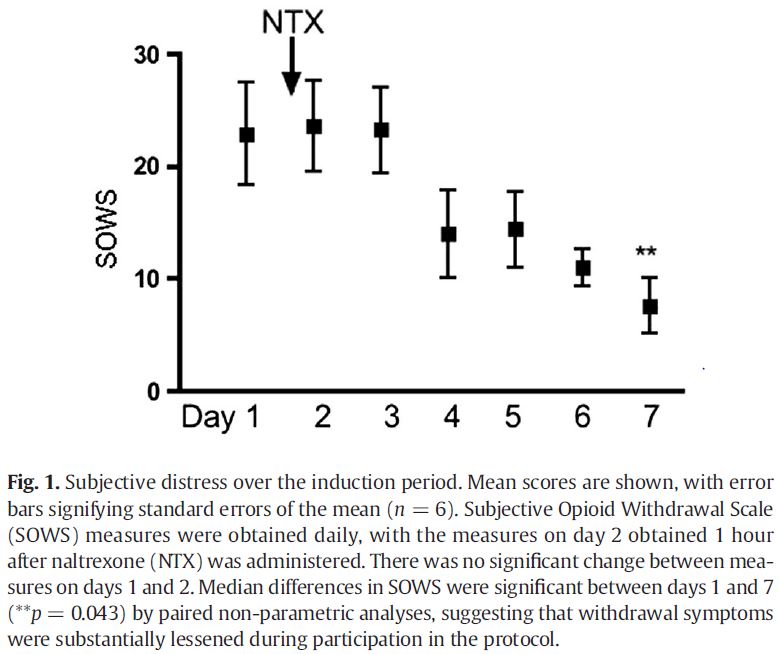
Participants (n = 6) took their last dose of buprenorphine the morning of admission for the inpatient phase of the study. The patients received an oral dose of naltrexone (6.25 mg) 48 hours after admission; the dose doubled each subsequent day until reaching a 50 mg dose. If able to tolerate this dose, patients received an intramuscular injection of 380 mg depot long-acting naltrexone which lasts for approximately one month. Medications to treat distress related to withdrawal symptoms (e.g., clonidine, clonazepam, ibuprofen) were available during this phase.
After administration of injection naltrexone, the patients returned for five weekly assessments which included 12-panel urine toxicology testing and administration of the Subjective Opioid Withdrawal Scale (SOWS), Spielberger State-Trait Anxiety Test, and Craving Scales.
Patients were mostly White (5 out of 6), male (4 out of 6), and employed (5 out of 6) with an average age of 40 years. The average dose of buprenorphine at the start of the study was 0.75 mg. All patients were previously unable to discontinue buprenorphine while under medical supervision; one participant had three previous failed attempts. The study procedure was tolerated by all participants with no adverse events.
While all participants experienced opioid withdrawal symptoms, clonazepam and clonidine were used in all participants to manage anxiety and restlessness; these medications were discontinued after naltrexone induction. While 2 of 6 patients reported sleep difficulties and 3 of 6 reported fatigue, all issues were resolved by the end of the 6-week trial. All participants self-reported abstinence from opioids (including buprenorphine) by 6-month follow-up.
Source: (Dakwar & Kleber, 2015)
IN CONTEXT
This open-label trial is the first step in determining the feasibility and tolerability of naltrexone-facilitated buprenorphine discontinuation prior to designing and recruiting for a larger trial.
Injection naltrexone was administered earlier than the current recommendation of 14 days after last opioid use and did not result in precipitated withdrawal or other adverse events, suggesting that naltrexone may be a good option for facilitating discontinuation of opioid agonists.
No patients required an additional injection of naltrexone after the first month allowing them to be free of all opioid treatment medications.
This method may also be used to facilitate a transition from treatment with a full opioid agonist (e.g., methadone) or partial agonist (e.g., buprenorphine) to treatment with an antagonist (e.g., naltrexone) if the patient is not ready to discontinue any kind of medication-assisted treatment.
Antagonists are beneficial because they do not produce dependence in the same manner as agonists and partial agonists. However, they do not provide the same protection against cravings and relapse. Thus, recently detoxified patients may not respond as well to antagonists initially and may need a method of facilitation (i.e., suboxone) to transition to their use.
Antagonists are beneficial because they do not produce dependence in the same manner as agonists and partial agonists. However, they do not provide the same protection against cravings and relapse. Thus, recently detoxified patients may not respond as well to antagonists initially and may need a method of facilitation (i.e., suboxone) to transition to their use.
While this study cannot establish efficacy of this method for either purpose, it is encouraging that all six patients remained opioid-free at 6-months especially since this was a group that was unable to stop using buprenorphine in previous attempts.
- LIMITATIONS
-
- It should be noted that other types of therapy (e.g., counseling or mutual help group attendance) used during this time period is unknown.
- Since this group was more stable in their recovery, this intervention may not work in a recently detoxified sample that has not had time to develop the skills needed to maintain their recovery.
NEXT STEPS
Future studies should investigate how long a patient should be maintained on buprenorphine before tapering off and discontinuing altogether. Additionally, more work is needed to increase feasibility of naltrexone induction since the current study required an inpatient stay and careful monitoring.
BOTTOM LINE
- For individuals & families seeking recovery: Medication-assisted treatment substantially lowers the risks associated with illicit opioid use (e.g., accidental overdose). Allow for the to development of recovery-oriented skills prior to medication discontinuation. When ready to discontinue use, consult an addiction-trained physician for assistance.
- For scientists: With this open-label trial as a starting point, researchers should test the efficacy of naltrexone-facilitated discontinuation and other methods through randomized controlled trials.
- For policy makers: Medication-assisted treatment is an important option for people suffering from opioid addiction. Given its risks, it may be beneficial for patients to taper off of these medications after achieving full sustained remission and adequate recovery capital (e.g., stable employment). More funding is needed to determine the best methods for doing so.
- For treatment professionals and treatment systems: While this trial suggests tolerability and feasibility of naltrexone-facilitated discontinuation of buprenorphine, it has not yet been proven to be effective. This kind of treatment requires coordination between different levels of care due to the transition from the inpatient induction period to weekly outpatient monitoring and the potential need for monthly naltrexone injections.
CITATIONS
Dakwar, E., & Kleber, H. D. (2015). Naltrexone-facilitated buprenorphine discontinuation: a feasibility trial. J Subst Abuse Treat, 53, 60-63. doi: 10.1016/j.jsat.2015.01.004

