High dose baclofen shows some promise in reducing high-risk alcohol use among primary care patients, but more research is needed
Few medications exist to help individuals reduce high-risk drinking. Low to moderate dose baclofen has been evaluated in recent years but has so far demonstrated minimal or no impact on drinking outcomes. While researchers in this study found that high-dose baclofen reduces high-risk drinking among primary care patients, some of their research design choices, as well as baclofen’s side effects, suggest further study is needed to arrive at a more precise estimate of benefits and for whom in particular the medication may be best suited.
WHAT PROBLEM DOES THIS STUDY ADDRESS?
High-risk drinking and alcohol use disorder remain prevalent, debilitating, and deadly, and few medications exist to help patients reduce and maintain recommended low-risk levels of drinking. Established medications for alcohol use disorder, such as naltrexone/vivitrol, work to block the reinforcing effects of alcohol, reduce cravings, or in the case of disulfiram (Antabuse), inhibit the alcohol metabolism process and increase physical discomfort after drinking. Baclofen, a muscle relaxant (GABA agonist), is a relatively new drug that has been evaluated previously but shown to have inconsistent effects on patients’ drinking, possibly due to low doses tested in previous studies, or reliance on testing the medication on individuals with more severe alcohol use disorder. This study addressed this prior limitation by evaluating high-dose baclofen, gradually increased over one year based on clinical judgment from patients’ care teams and its side effects among patients drinking above recommended limits within primary care settings.
HOW WAS THIS STUDY CONDUCTED?
This was a large double-blind randomized clinical trial (RCT) comparing baclofen to placebo on drinking outcomes over 12 months. Participants included 320 patients (70% men) across 62 primary care clinics in France. All participants were between the ages of 18 and 65, had never taken baclofen, and were not currently taking other medications to help individuals reduce or quit drinking, such as acamprosate, naltrexone, or disulfiram. All participants were also engaged in “high-risk” alcohol use during the three months before entering the study, which was defined as women drinking more than 40g of ethanol – approximately three U.S. standard drinks per day – and for men, drinking more than 60g of ethanol – just over four U.S. standard drinks per day. Once these treatment seeking participants were consented, they were randomized to receive baclofen or a placebo, beginning at doses of 15mg/day for the first three days, then increasing doses up to, but often not reaching, 300mg/day in consultation with their prescribing physician. In cases where the patient believed the medication to which they were assigned was ineffective, they could opt for “open-label” use, which meant that the intervention was no longer masked and any/all participants who requested baclofen could use it. These individuals essentially opted out of the trial and were considered “treatment failures” for the purposes of the primary statistical analyses – that is, the researchers considered them treatment failures to examine only outcomes among the participants who remained naïve to the study condition to which they were originally assigned. By the end, the median daily doses of medication reached were 180mg and 210mg per day in the baclofen and placebo groups, respectively.
The researchers were primarily interested in assessing the rates of achieving “low-risk” drinking (20g/40g [roughly 1.5/3 U.S. standard drinks] or less per day for women/men, respectively), using daily diary self-reports returned during in-office visits, by the last month of the 12-month trial among patients who did not switch to “open-label” baclofen use.
They also examined success rates among participants between treatment groups regardless of whether they switched to “open-label” baclofen use. The researchers also collected information from patients regarding average daily drinking, number of days abstinent or spent engaged in heavy drinking via daily diaries collected by the primary care physicians. Participants completed measures of depression, obsessive-compulsive drinking, and quality of life at baseline and months three, six, and 12. The researchers also compared the side effect profiles of patients between groups.
There were 320 primary care patients recruited and randomized in the current study (baclofen = 162, placebo = 158). Patients were 46.5 years old on average, predominantly male (70%), employed (58%), consuming alcohol daily (84%), and met criteria for DSM-IV alcohol dependence (94.5%).
WHAT DID THIS STUDY FIND?
Patients who received baclofen were more likely to achieve “low-risk” drinking relative to patients who received placebo, but only when individuals who voluntarily switched to open label were considered treatment failures.
When individuals who voluntarily switched to open label baclofen were considered “treatment failures”, and could not by definition be included in the analysis as having achieved “low-risk” drinking, 57% in the baclofen group achieved “low-risk” drinking versus 36% in the placebo group. This effect was statistically significant indicating a difference of 21%.
However, 12% of the baclofen group and 38% of the placebo group switched to open-label baclofen. Thus, when individuals who switched to “open-label” baclofen (i.e., unmasked use) were not automatically considered “treatment failures”, the difference between groups was no longer significant. That is, placebo participants had similar rates of “low-risk” drinking compared to baclofen participants, on average.
For other drinking, as well as functioning outcomes, baclofen and placebo generally had similar outcomes.
Baclofen participants had more alcohol abstinence days, on average, compared to placebo during the last month of the 12-month trial (i.e., 15 vs. 11 abstinence days), and lower mean alcohol consumption (10g less on average) between months 6 and 12. However, the groups had similar heavy drinking days (defined as > 40/60 grams of pure ethanol per day for women/men, respectively), likelihood of meeting diagnostic criteria for alcohol dependence (based on DSM IV), alcohol craving, anxiety, and functioning.
Adverse events were more common among patients who received baclofen.
Drowsiness, fatigue, and insomnia were the most frequently reported side effects in this study. Serious adverse effects were also more likely among individuals receiving baclofen (38% of patients) vs. placebo (23% of patients). Seven deaths occurred in the baclofen group, three in the placebo group, but none were attributed to the study medication. Three patients who received baclofen and zero who received placebo experienced a manic episode.
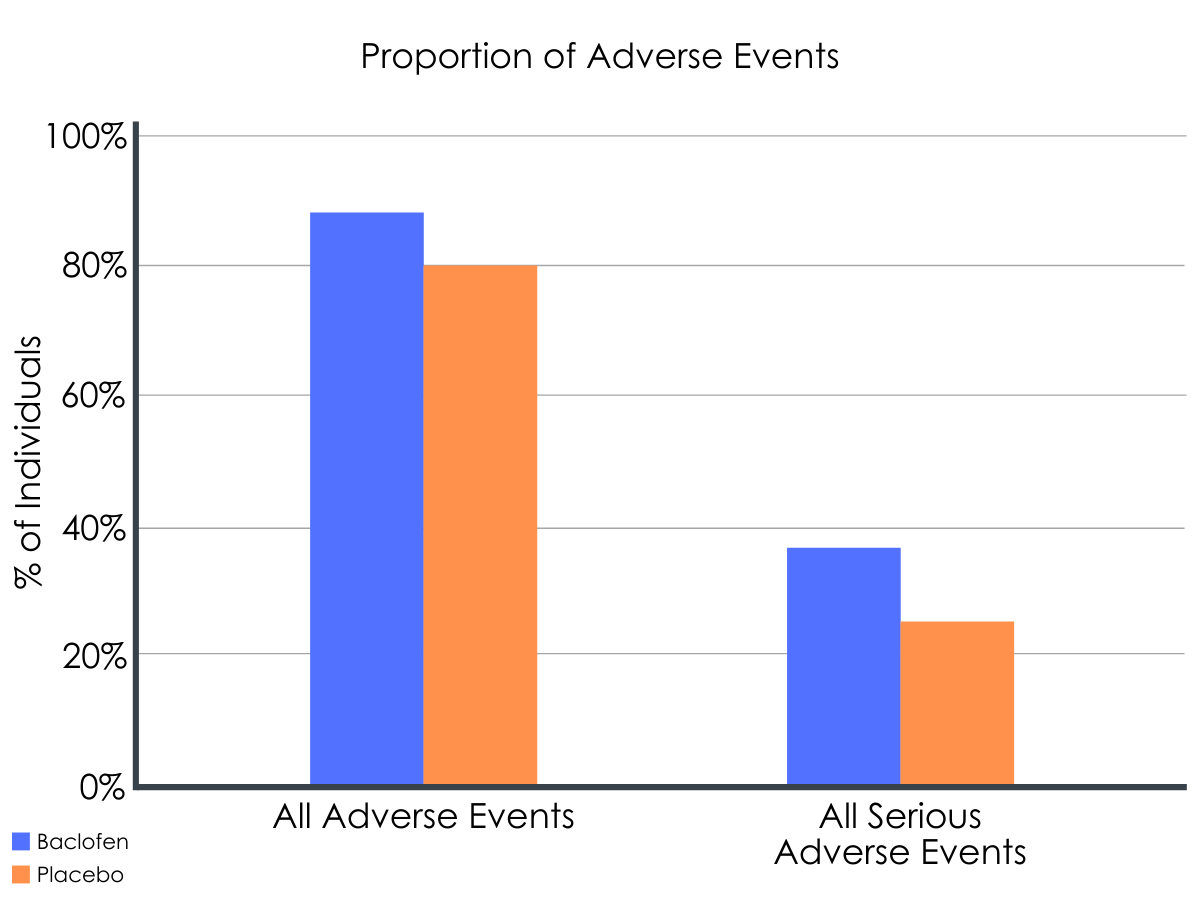
Figure 1. Percentage of patients experiencing adverse events correspond to numbers of patients with at least one occurrence of each adverse event.
Adverse events include drowsiness, fatigue, insomnia, cognitive function disorders, sweat, vertigo, hypothymia, nausea, paresthesia, memory disorders, headache, tinnitus, sexual disorders, agitation, anxiety disorders, vomiting, psychiatric disorders excluding dysthymia, fall, tremor, hot flush, muscular spasm, diarrhea, decreased appetite, and hyperthymia.
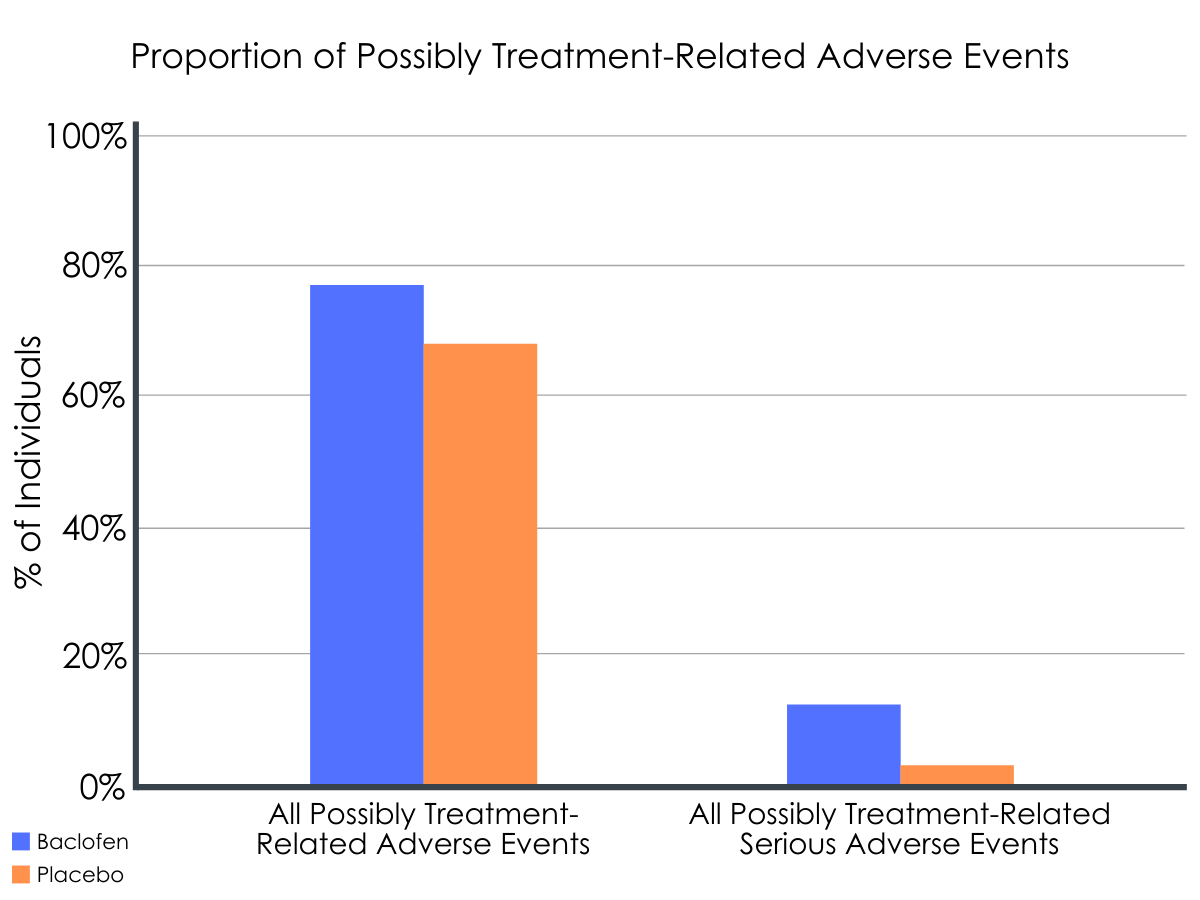
Figure 2.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?
Patients who received baclofen were more likely to achieve “low-risk” drinking by the 12th month and reported less daily drinking and more abstinence days compared to patients who received placebo. However, the difference between groups was non-significant when patients who transitioned to “open-label” baclofen use (19 vs. 60 patients in the baclofen vs. placebo group, respectively) were not considered “treatment failures.” One explanation for this might be that the use of baclofen among the 60 participants in the placebo condition “made up” some of the difference that would otherwise exist between groups if placebo patients were not allowed to switch to “open-label” baclofen.
On the other hand, it is also possible that participants in the placebo condition were aware they received a placebo given the relative lack of side effects and because they were drinking more heavily. This could have prompted them to ask for baclofen directly and thus switch to “open-label” use, resulting in treatment “failure,” and an overestimation of the effect of baclofen in the primary analysis. Unfortunately, it is not possible to know which is true in the current study, or whether outcomes would have been different if patients were not allowed to switch to “open-label” medication use, as is more typical in randomized studies of medication.
At the same time, adverse and serious adverse events were more likely among patients who received baclofen and should be taken seriously in future studies. In particular, baclofen participants had more serious adverse events, and more instances of drowsiness overall, relative to placebo. The researchers also note that side effects were more severe in this study of current drinkers compared to other baclofen studies that tend to recruit recently detoxed and/or abstinent patients, and attribute this to possible interactions between alcohol and baclofen in the current study.
Therefore, baclofen potentially may be helpful in treating harmful drinking and alcohol dependence in primary care patients. But given the current study’s design limits, as well as the somewhat risky side effect profile of baclofen, this study, overall, does not yet indicate baclofen as a front-line treatment for individuals with alcohol dependence in primary care. Rather, significantly more research is needed to evaluate the efficacy of baclofen against other established FDA-approved medications that have shown reliable albeit modest benefits among patients with alcohol use disorder. Comparative clinical trials are also needed to evaluate the side effect profiles of baclofen vs. other FDA-approved medications for alcohol use disorder (e.g., naltrexone, disulfiram, acamprosate).
- LIMITATIONS
-
- Significantly more patients discontinued medication in the placebo group vs. baclofen group. The fact that a significantly greater proportion of placebo participants switched to open-label use is by itself a significant research limitation, but also leaves open the possibility that others in the placebo group were aware of their group assignment, which could have also biased results. Given the more pronounced side effect profile of baclofen (e.g., marked drowsiness, fatigue, etc.), this may be quite likely.
- Fewer placebo patients (34%) completed their final 12-month diary compared to baclofen patients (42%), which might also bias results.
- The switch to “open-label” baclofen use was a planned modification to prevent patients from leaving the study and securing baclofen elsewhere. However, switching and officially unmasking medication use in the current study undermines interpretability of the findings.
- The inclusion/exclusion criteria (e.g., exclusion of court-ordered patients and those without health insurance) limits generalizability of the findings.
- The researchers do not provide information about other interventions sought or received by patients in the current study, which could include mutual/self-help (e.g., Alcoholics Anonymous), outpatient treatment, or residential care. They do report the number of patients hospitalized and admitted to detox but not proportion in each treatment group.
BOTTOM LINE
- For individuals and families seeking recovery: Patients treated with baclofen were more likely to reduce their drinking to “low-risk” levels over one year, drank less per day, and reported more abstinent days per month compared to placebo control patients. The side effects were also more common among those treated with baclofen. In particular, individuals with bipolar disorder may want to carefully consider use of baclofen, as 3 patients treated with baclofen in the current study experience a manic episode (but none in the placebo condition). For the most part, the side effects attributable to the medication were minor (e.g., drowsiness, fatigue, insomnia). While the mechanisms of action are uncertain, baclofen has been shown to help individuals feel more indifferent toward alcohol. Keeping in mind that much more work is needed to evaluate the effectiveness of baclofen, especially given the limitations of the current study, baclofen might be an appealing medication for some individuals trying to cut back or abstain from drinking among patients who have already tried other medications, such as disulfiram (e.g., Antabuse), naltrexone (e.g., ReVia, Vivitrol), and acamprosate (e.g., Campral).
- For treatment professionals and treatment systems: In this study, patients assigned to receive baclofen were more likely to achieve “low-risk” drinking by after 12 months, and reported fewer drinking days and drinks per drinking day compared to placebo patients. Taking into account the side effects observed, baclofen may represent another medication option for some individuals interested in cutting back or abstaining from alcohol use, and has been shown to compare favorably to other medications (e.g., naltrexone, acamprosate). Unlike previous baclofen studies, the patients in this study were not abstinent nor were they selected following completion of a detox program. Thus, the current study demonstrates that baclofen was reasonably well-tolerated among active drinkers who are trying to stop or cut down on their drinking. However, given the noted limitations of this study, more research is needed to evaluate the effectiveness of baclofen, especially relative to other front-line FDA-approved medications (e.g., naltrexone, acamprosate). This is significant and in line with reports that moderation is a reasonable alternative to abstinence only for some, and that even modest reductions in alcohol consumption can lead to significant long-term health benefits.
- For scientists: In a large-scale double-blind placebo-controlled trial spanning 62 primary care clinics in France, patients assigned to receive baclofen were more likely to achieve “low-risk” drinking after 12 months compared to placebo patients. Baclofen patients also reported lower average drinking and reported an average of 3 more abstinence days per month. However, the difference between conditions disappeared once researchers reclassified “open-label” baclofen users in both groups. This may have been due to the high proportion of placebo patients receiving “open-label” baclofen and benefiting from the treatment. Alternatively, placebo patients may have surmised they were in the placebo condition due to lack of noticeable side effects, which were more frequent in the baclofen condition, and left the study or opted for “open-label” baclofen, thereby becoming treatment “failures.” If so, this could have led to an overestimation of baclofen efficacy in the primary analysis. Replication is needed to evaluate these possibilities. In addition, the researchers’ design allowed for evaluation of baclofen under “real-world” conditions and using an intent-to-treat analysis. At the same time, evaluation of differential effects based on dose may be important in future studies, particularly given the dearth of research on high-dose baclofen for alcohol use disorder. Finally, significantly more research is needed to examine both the effectiveness and side effect profile of baclofen relative to other FDA-approved medications for alcohol use disorder.
- For policy makers: This French study provides some support for the use of baclofen to treat high-risk drinking and alcohol use disorder, as evidenced by higher rates of “low-risk” drinking among primary care patients treated with baclofen vs. placebo. Execution of this project required coordination with providers from across 62 primary care clinics, as well as significant research funding. While these results are promising, it is also clear that more work is needed to identify how baclofen works, for whom, to identify optimal doses, and to evaluate its effectiveness and tolerability relative to other FDA-approved medications. To accomplish this, increased funding and coordination across large healthcare systems would help scientists continue evaluating medication outcomes in “real-world” settings.
CITATIONS
Rigal, L., Sidorkiewicz, S., Tréluyer, J. M., Perrodeau, E., Le Jeunne, C., Porcher, R., & Jaury, P. (2019). Titrated baclofen for high-risk alcohol consumption: A randomized placebo-controlled trial in out-patients with 1-year follow-up. Addiction, 115(7), 1265-1276. doi: 10.1111/add.14927

