The CBD panacea? The jury is still out on whether CBD can help treat cannabis use disorder
As policies on the production and sale of cannabis become more liberalized across the United States and internationally, increasing numbers of individuals have begun to consume it. Given that cannabis exposure can result in a cannabis use disorder for some, more people are likely to develop cannabis use disorders and, in fact, rates of such disorders have increased in the population. Currently, there are no proven pharmacotherapies to treat cannabis use disorder. A question has lingered, however, as to whether certain cannabis plant derivatives – notably cannabidiol (more commonly known as CBD) – often publicized as a treatment for a wide variety of medical conditions, could be effective in treating cannabis use disorder. This rigorous clinical trial addressed this question.
WHAT PROBLEM DOES THIS STUDY ADDRESS?
Cannabis is one of the most commonly used psychoactive substances worldwide, and cannabis use disorder is the most prevalent type of drug use disorder in the United States (other than alcohol). The potency of cannabis products has increased, nearly doubling in the United States over the past 10 years, with higher potency associated with earlier onset and greater severity of cannabis use disorder. It has become increasingly important to develop novel treatments for cannabis use disorder, particularly as more countries and regions move toward cannabis legalization and people have easier access, which will likely lead to more cannabis use and increased cannabis addiction.
Cannabidiol (CBD) has emerged as a possible treatment for a wide range of medical conditions, oftentimes being touted as a valid treatment for some of these conditions without substantial evidence. Importantly, however, CBD has shown promise in reducing drug craving and anxiety in patients recovering from heroin use disorder, and may have some effectiveness as an adjunct treatment for some mental health conditions. CBD itself is a non-intoxicating cannabinoid derived from cannabis. Particularly in light of the evidence for opioid agonist medicines in the treatment of opioid use disorder, it seems very reasonable to explore this medication as a treatment for cannabis use disorder, as no FDA-approved pharmacotherapy currently exists and any positive findings could increase treatment options for a highly prevalent condition and lead to the first empirically supported pharmacotherapy for cannabis use disorder. In this study, researchers examined the effect of CBD for the treatment of cannabis use disorder using a rigorous research design.
HOW WAS THIS STUDY CONDUCTED?
This study used a randomized clinical trial design with 84 participants who received one of three different CBD medication doses or a placebo twice daily for four weeks and were assessed for cannabis use using both biological and self-report measures weekly – urinary tetrahydrocannabinol (THC) levels and number of days per week abstinent from cannabis.
This was a phase 2a clinical trial, meaning that a small sample size was used to determine the most efficacious dose of CBD for treating cannabis use disorder, which may inform a larger clinical trial that tests the best dose determined by this initial trial. The researchers used a design technique to increase the efficiency of the study (called an adaptive Bayesian trial). That is, 48 participants were initially randomized to receive 200mg of CBD (n=12), 400mg of CBD (n=12), 800mg of CBD (n=12), or placebo (n=12) for four weeks. Interim analysis showed 200mg of CBD to not be an efficacious result, so the second stage of the trial randomized 34 different participants to 400mg of CBD (n=12), 800mg of CBD (n=11), and placebo (n=11) for four weeks. Study participants from both stages were added to the final analysis with the exception of the group that received 200mg of CBD. Notably, all groups including the placebo also received 30 minutes of motivational interviewing targeting cannabis use change from a psychologist at the screening visit, baseline visit, and weekly visits during the study period (six times over a four-week period).
This study was done in London, United Kingdom and participants were recruited through websites, forums, and flyers in the local community. The first stage was done between 2014-2015 and the second stage was done between 2016-2017. Inclusion criteria to be a part of the study included: aged 16-60 years old, met Diagnostic Statistical Manual (DSM-5) criteria for cannabis use disorder of at least moderate severity, expressed a desire to stop using cannabis and intended to do so in the next month, reported at least one previous unsuccessful attempt to quit cannabis, reported co-administering cannabis and tobacco together (common in Europe), and provided a positive urine sample for THC. Some of the exclusion criteria included: currently pregnant or breastfeeding, currently using a psychotropic drug to treat a mental illness, and the use of other illicit drugs more than twice per month at screening. The study used a synthetic, pharmaceutical-grade form of CBD of 99.9% purity that was delivered in a capsule.
In addition to the weekly site visits during the four weeks of the clinical trial when participants received the CBD (or placebo), there were follow up points after the study at 6 weeks (site visit), 8 weeks (telephone), 12 weeks (site visit), 16 weeks (site visit), 20 weeks (telephone), and 24 weeks (telephone). In addition to the biological and self-reported primary outcomes measured, secondary outcomes measured included cannabis withdrawal, alcohol and tobacco use, sleep quality, and depression and anxiety symptoms. Being a clinical trial to partly assess the safety of CBD, adverse events were also recorded.
In order to ensure adherence to the treatment plan, study participants were sent text messages to remind them to take their medication two times a day, spaced 12 hours apart. If participants were not adherent to the treatment protocol, determined by more than 30% of self-reported doses missed per week, returning more than 30% of medication capsules per week, beginning psychotropic medication during the study period, or not attending a site visit within two days of the scheduled appointment, they were not provided with additional medication for the duration of the trial but still received motivational interviewing. Overall, 77 or the 82 participants, or 94.2%, adhered to the treatment protocol and completed treatment.
This clinical trial’s data were analyzed using an “intention-to-treat” approach, meaning that even if participants dropped out of the study or were determined to not adhere to the treatment protocol, they were still analyzed as part of the group to which they were initially randomly assigned.
A complex statistical technique was used after the first and second stage of the adaptive Bayesian trial to determine the most efficacious dose of CBD compared with the placebo. The treatment effect of CBD was determined by comparing the change in baselines of the primary outcome measures, urinary THC levels (specifically a urinary THC-COOH:creatinine ratio) and self-reported days of cannabis abstinence per week, of the 400mg and 800mg CBD groups to the placebo group.
The group that received 400mg of CBD (n=24) and 800mg of CBD (n=23) were similar to the placebo group (n=23) in both age (26.58 vs. 27.43 vs. 24.87 years old, respectively) and sex (71% vs. 70% vs. 74%, respectively). The group that received 400mg of CBD may have had a higher severity of cannabis use disorder, as measured by an average of 9.0 DSM-V cannabis use disorder symptoms for these participants compared with an average of 8.5 symptoms for the 800mg group and an average of 8.6 symptoms for the placebo group.
WHAT DID THIS STUDY FIND?
The 400mg and 800mg dose of CBD, but not 200 mg, outperformed placebo in reducing cannabis use over the study period.
The 200mg dose of CBD was determined not to be an efficacious dose to reduce cannabis use in those with cannabis use disorder. Compared with placebo, the group taking 400mg of CBD had decreased urinary THC levels by 94.2 ng/ml and reported increased abstinence from cannabis by 0.5 days per week during the four-week treatment period whereas the group taking 800mg of CBD had decreased urinary THC levels by 72.0 ng/ml and reported increased abstinence from cannabis by 0.3 days per week (although this latter measurement was not statistically significant). At final follow up (24 weeks), the group on 400mg of CBD sustained their cannabis reduction compared with the placebo group whereas the group on 800mg of CBD had similar cannabis use outcomes to the placebo group.
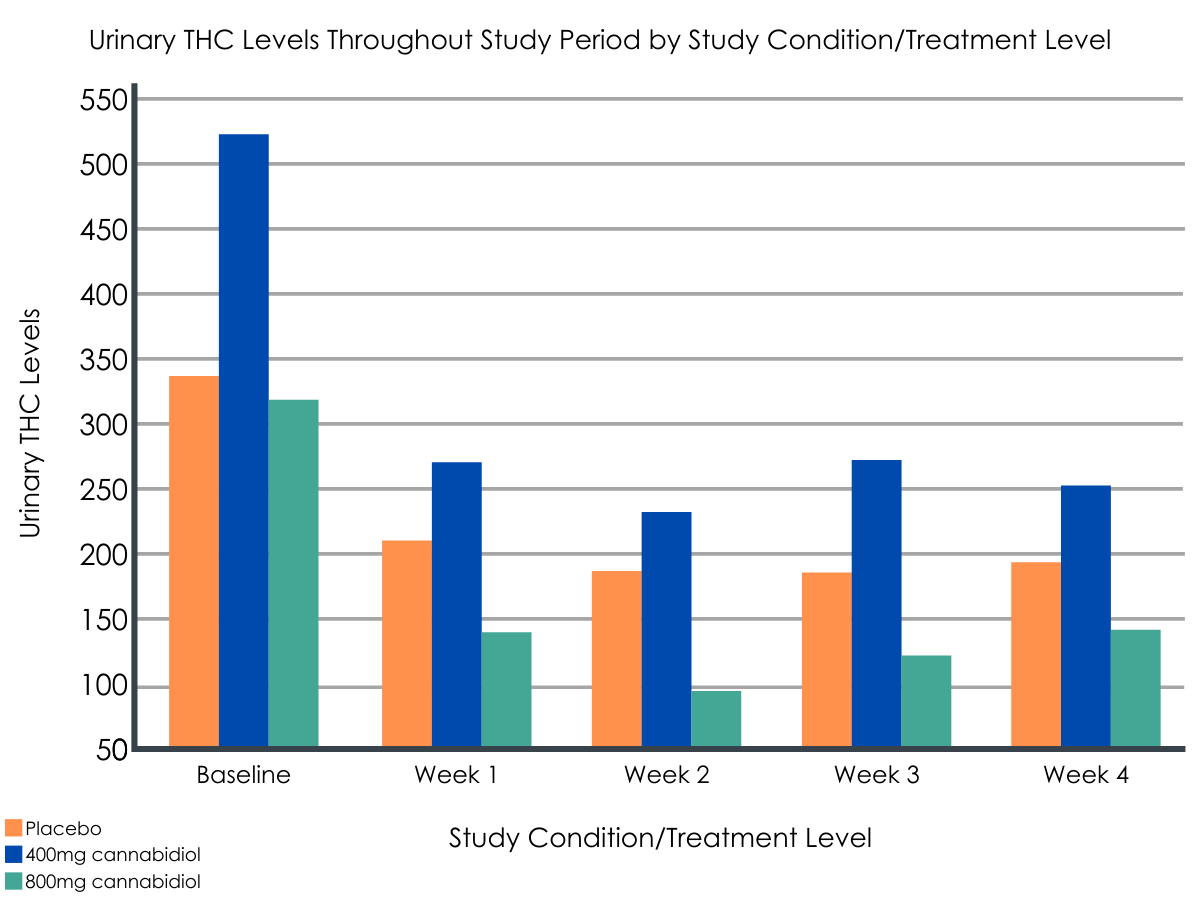
Figure 1.
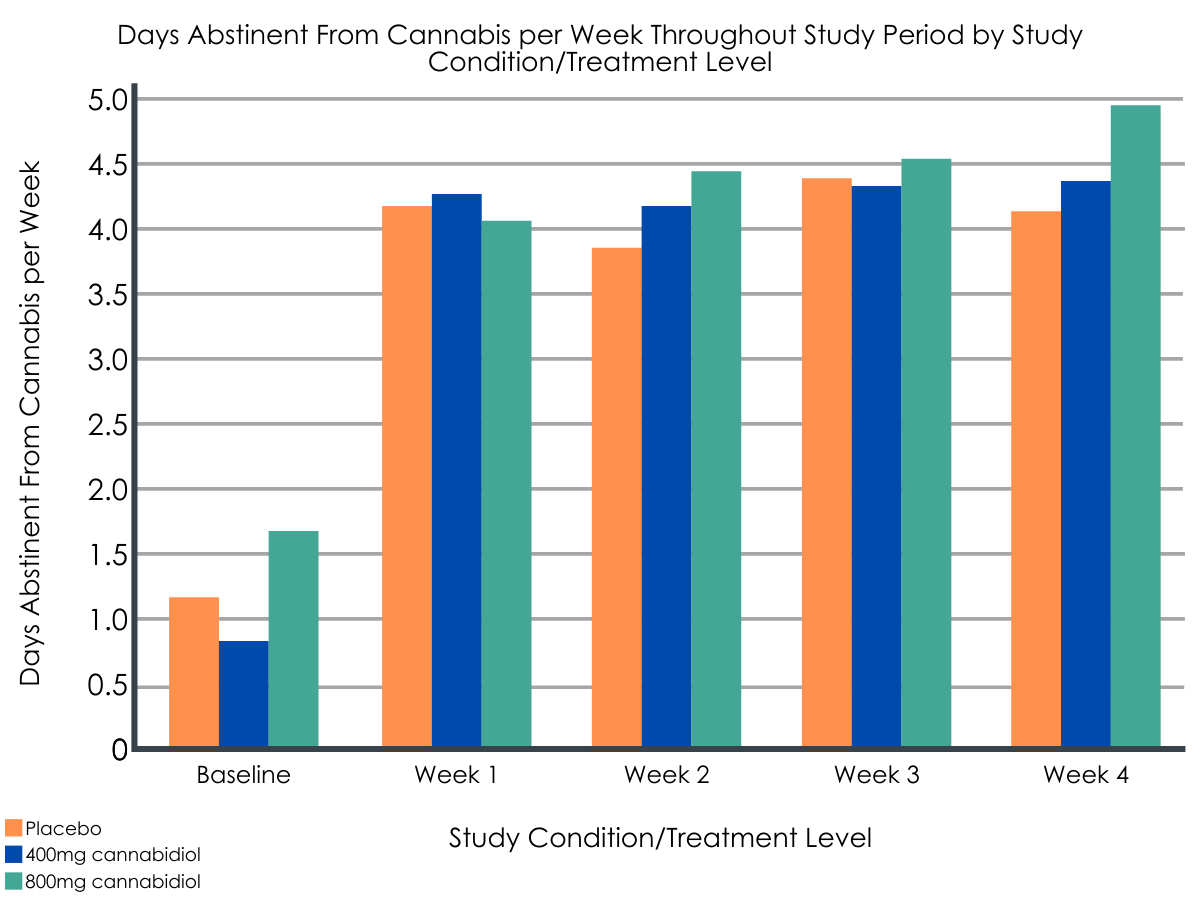
Figure 2.
The 400mg and 800mg dose of CBD were generally well-tolerated by study participants.
The number of mild and moderate adverse events reported by the study participants did not differ between the placebo group and each of the three groups taking CBD. No severe adverse events were reported and none of the participants dropped out of the study because of the treatment.
The CBD treatment appeared to have an effect on cigarette smoking, sleep quality, and both cannabis withdrawal and anxiety symptoms.
The group taking 400mg of CBD decreased the number of cigarettes smoked and had a decrease in sleep quality during the treatment period and at final follow-up. The group taking the 800mg dose of CBD reported reduced cannabis withdrawal symptoms and reduced anxiety symptoms during the treatment period and at final follow-up.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?
In a group of 82 patients with moderate to severe cannabis use, daily doses of 400mg and 800mg of CBD over a four-week period were found to result in a small but significant reduction in cannabis use compared to placebo, although once the medication was discontinued, this reduction was only sustained by the group taking 400mg of CBD at final follow-up 24 weeks later.
Current evidence-based treatment for cannabis use disorder is limited to behavioral therapy, including motivational enhancement therapy, contingency management, and cognitive-behavioral therapy. Although several types of pharmacotherapies have been investigated, there is not sufficient evidence to date demonstrating a benefit for any of these pharmacotherapies. Additionally, most people who report resolving a cannabis problem do not use formal treatment and less than 10% of those with a cannabis use disorder received cannabis-specific treatment, highlighting that only a small minority of people with more severe variants of cannabis use disorder would likely benefit from a new pharmacotherapy. However, the availability of pharmacological options for cannabis use disorder may make treatment more attractive, and therefore increase treatment engagement for individuals with cannabis use disorder.
Importantly, given the rhetoric that CBD can treat a wide range of medical conditions without being backed by substantial evidence to date, the researchers’ effort to apply scientific rigor to the study of CBD for cannabis use disorder is commendable. However, more research is needed before CBD should be considered as a treatment option for cannabis use disorder.
Although this study established that CBD is generally safe in the treatment of cannabis use disorder, more research is needed to draw conclusions about its effectiveness. In addition to the treatment effect being small during the four-week trial, only the group taking the 400mg dose had sustained a reduction in cannabis use at the final follow-up. Moreover, even though the severity of cannabis use disorder in the different groups was similar as measured by DSM-V symptoms, the baseline measures for the group taking the 400mg dose of CBD had much higher urinary THC levels and reported less days abstinent from cannabis per week compared with placebo. Therefore, there is larger room for improvement in the group taking 400mg of CBD which can lead to a regression to the mean, sometimes a problem in small sample sizes where randomization does not even out pre-existing individual differences in each treatment group. If future trials with larger sample sizes do explore the use of CBD to treat cannabis use disorder, this study established that the likely efficacious dose is between 400mg and 800mg. Importantly, the study used a synthetic, pharmaceutical-grade form of CBD rather than a plant extract and the daily doses of CBD used (200-800mg) are much larger than the concentrations typically found in unregulated over-the-counter CBD products (25-50mg).
The placebo group also improved over the four-week treatment period. The placebo group went from having a little more than one day of abstinence per week at baseline to around four days of abstinence per week at the end of the treatment period, and urinary THC levels also decreased. All groups in this clinical trial received six sessions of motivational interviewing, an evidence-based intervention for cannabis use disorder. This behavioral intervention appears to have been effective in reducing cannabis use and may have had more of an impact than the CBD, though we cannot be sure due to the likely clinical effect of the placebo medication.
There were some interesting findings of the effect of CBD on some of the secondary outcomes in this study. The 400mg dose appeared to decrease the number of cigarettes smoked by participants during the treatment period and at the final follow-up. This finding may warrant further investigation given the increased tobacco-related mortality in those with substance use disorders. The 800mg dose appeared to moderately alleviate cannabis withdrawal symptoms and anxiety symptoms during the treatment period and at final follow-up, opening up the possibility that CBD could be used to treat symptoms in those seeking treatment for cannabis use disorder that may improve retention in treatment and other treatment outcomes.
Although this study showed that CBD doses ranging from 400mg to 800mg have the potential to reduce cannabis use in those with moderate or severe cannabis use disorder, taking into account the marginal effect size, the small sample size, and other limitations of the study (see limitations section below), these study results should be interpreted with caution and more research is needed before making a clinical recommendation of using CBD to treat cannabis use disorder.
- LIMITATIONS
-
- This randomized clinical trial, although of rigorous design, had a small sample size and used more liberal probability cutoffs of 0.9, meaning that there is a 10% chance that the treatment effect of CBD seen in this study was a result of random error.
- Adherence to the treatment protocol was essentially measured by self-reported measures whereas many clinical trials deliver medications at site visits.
- The baseline measurements of urinary THC level and days of abstinence from cannabis for the 400mg CBD group and the placebo group were substantially different although the two groups were similar in the severity of cannabis use disorder as measured by DSM-V symptoms. Regression to the mean and THC metabolism may play a big role in determining the treatment effect of CBD in the 400mg group due to the small sample size and the inability to even out pre-existing differences and their potential for confounding true treatment effects among participants in the different groups.
- The study period was only four weeks and the bioavailability of CBD can vary based on food consumption, limiting the long-term treatment effect and efficacious dose of CBD respectively.
- This study took place in the United Kingdom and used participants that co-administered cannabis and tobacco together, did not use psychotropic medications for mental health conditions, and were not frequent polysubstance users, which may not represent the large majority of people with cannabis use disorder.
- Patients received daily text reminders to increase treatment adherence during the clinical trial. Although providers could theoretically implement such a strategy, this is not yet common in real-world practice. Thus, the marginal effect of CBD should be considered in context of the enhanced treatment support that participants received.
BOTTOM LINE
In a group of 82 patients with moderate to severe cannabis use disorder, daily doses of 400mg and 800mg of CBD over a four-week period were found to result in a small but significant reduction in cannabis use compared to placebo when combined with six sessions of behavioral intervention, although this reduction was only sustained by the group taking 400mg of CBD at final follow-up 24 weeks later. The jury is still out in determining if CBD is an effective medication for treating cannabis use disorder. However, there are effective treatment modalities to treat cannabis use disorder, including motivational enhancement therapy, contingency management, and cognitive-behavioral therapy.
- For individuals and families seeking recovery: More research is needed to confirm the extent to which CBD is an effective medication treatment for those with moderate or severe cannabis use disorder. However, there are effective treatment modalities to treat cannabis use disorder, including motivational enhancement therapy, contingency management, and cognitive-behavioral therapy.
- For treatment professionals and treatment systems: The jury is still out in determining if CBD is an effective medication for treating cannabis use disorder. However, there are other treatment modalities to treat cannabis use disorder that are evidence-based, which include behavioral therapies such as motivational enhancement therapy, contingency management, and cognitive-behavioral therapy.
- For scientists: In a double-blind, placebo-controlled, randomized clinical trial using an adaptive Bayesian design, a group of 82 patients with moderate to severe cannabis use disorder taking daily doses of 400mg and 800mg of CBD over a four-week period were found to have a small but significant reduction in cannabis use compared to placebo, although this reduction was only sustained by the group taking 400mg of CBD at final follow-up 24 weeks later. The group taking the 400mg dose decreased cigarette use and the group taking the 800mg dose reported decreased cannabis withdrawal and anxiety symptoms. Given the small sample size and other limitations of this study, further research is needed to confirm these findings, which may be a fruitful endeavor given the high prevalence of cannabis use disorder and the absence of proven pharmacotherapies. This was a phase 2a clinical trial that did not progress to a phase 2b trial due to lack of funding, so there is an opportunity to build on this existing work.
- For policy makers: The jury is still out in determining if CBD is an effective medication for treating cannabis use disorder. Given the rhetoric behind CBD being touted as a treatment for a wide range of medical conditions, funding rigorous research to examine these claims will create more consumer transparency and marketing accountability. In the context of CBD as a treatment for cannabis use disorder, further research may be a fruitful endeavor given the high prevalence of cannabis use disorder and the absence of proven pharmacotherapies.
CITATIONS
Freeman, T. P., Hindocha, C., Baio, G., Shaban, N. D. C., Thomas, E. M., Astbury, D., . . . Curran, H. V. (2020). Cannabidiol for the treatment of cannabis use disorder: a phase 2a, double-blind, placebo-controlled, randomised, adaptive Bayesian trial. Lancet Psychiatry, 7(10), 865-874. doi: 10.1016/S2215-0366(20)30290-X

