Does Vivitrol Work Equally Well for Different Types of Individuals with Opioid Use Disorder?
Once a month injectable naltrexone (brand name Vivitrol) has emerged as an evidence-based medication for opioid use disorder that has appealing qualities including no daily dosing requirements, no agonist properties that can induce drowsiness in some, and potentially reduced stigma. This study sought to determine if the medication worked equally across groups categorized by different demographics, addiction, mental health, and environment.
WHAT PROBLEM DOES THIS STUDY ADDRESS?
The opioid epidemic has resulted in hundreds of thousands of preventable deaths. Opioid use disorder affects all walks of life including those with varying levels of depression, socialization, risk for drug use, medical histories, employment, legal, and family issues. Previous research had reported Vivitrol can decrease relapse rates by about 10 to 20%, and that Vivitrol worked equally well to promote abstinence from opioids across a range of demographic and severity characteristics when tested against placebo in a Russian sample. This study investigated a similar question in a criminal justice sample while testing additional patient characteristics that could have implications for treatment matching.
HOW WAS THIS STUDY CONDUCTED?
The authors conducted a secondary data analysis of a randomized clinical trial at five sites throughout the United States comparing Vivitrol to counseling and referral to community resources, including methadone, buprenorphine, and treatment programs (i.e., treatment as usual).
- MORE ON STUDY METHODS
-
Participants included 308 community-dwelling adult volunteers with a history of both criminal justice involvement (e.g., on parole) and opioid dependence based on the fourth edition of the diagnostic and statistical manual of mental disorders (DSM). Recruitment did not occur directly from criminal justice authorities in order to minimize potential for coercion, rather, recruitment efforts were focused on medical clinics, addiction treatment programs, at-risk criminal justice populations, with additional efforts in print, radio, and online. About half of the participants (n=153) received an initial injection of 380mg depot Vivitrol followed by subsequent injections every four weeks during the 6-month treatment phase. Follow-up visits occurred over the course of 12 months.
For this analysis, the primary outcome was opioid relapse status, with relapse being defined as two or more consecutive positive or missing urine tests for opioids and/or 10 or more days of self-reported opioid use in any 28 day period.
The patient characteristics that were tested to see if they influenced the degree of medication benefit (i.e., statistically referred to as moderator effects) included baseline scores for depression, socialization, drug use risk, medical, psychiatric and employment status, alcohol use, legal and family-social issues.
WHAT DID THIS STUDY FIND?
The only significant characteristic that appeared to moderate the effects of Vivitrol on relapse was “drank alcohol to intoxication” in the 30 days prior to randomization. Those who reported recent intoxication and were assigned to Vivitrol relapsed to opioids at a rate (56%) similar to treatment as usual (58%), while those without recent intoxication had a lower rate of opioid relapse (41%) versus treatment as usual (65%).
The majority of patient characteristics tested did not appear to influence the effectiveness of Vivitrol. This means that overall, in a sample of individuals with a criminal justice history and living in the community, Vivitrol appears to work equally well across subgroups with diverse demographics, addiction histories, mental health, and environmental characteristics.
FIGURE BELOW: Patient characteristics tested to moderate (i.e., change) the effectiveness of Vivitrol. Blue dots towards the left favor the Vivitrol condition and towards the right favor referral to community resources (i.e., treatment as usual).
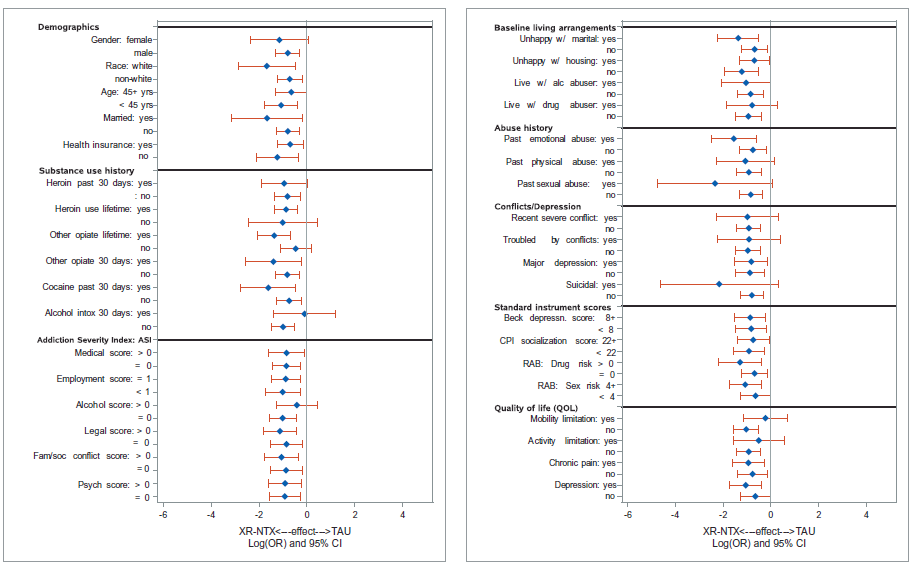
The figure above displays the likelihood of relapse (i.e., odds ratios) by the Vivitrol versus treatment as usual group, broken down by gender, race, age, marital status, and other characteristics. The likelihood of relapse is portrayed by the blue dot and the red line indicates the degree of confidence in the range of that estimate (i.e., confidence intervals). If the confidence intervals cross zero than there is no difference between Vivitrol and treatment as usual.
Closer inspection of the group who reported “yes” to alcohol intoxication in the past 30 days shows the blue dot is very close to zero, which means these participants who received Vivitrol are no more or less likely to relapse than the participant who received treatment as usual, while for those who reported “no”, they did better on Vivitrol compared to treatment as usual. Overall, the majority of these subgroup analyses favored Vivitrol so the different indicating that these different subgroups are equally likely to benefit from Vivitrol, which in turn was better rather than treatment as usual.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?
Health practitioners, researchers, and patients are increasingly interested in the potential of precision medicine, which implies the ability to identify which patients are the best candidates for which treatments. Moderator analyses were used to tests various patient characteristics to determine if there is a specific group for whom the medication works better or worse. In this case, the benefit for Vivitrol on reducing relapse to opioid use was reduced when patients reported alcohol intoxication within the past 30 days at treatment intake.
Given that patients need to be abstinent from alcohol before given Vivitrol, it is possible that current drinking does something to mute the medication’s mechanism of action and these individuals may have continued to consume alcohol heavily over time continuing to weaken its therapeutic effects. By identifying this patient characteristic, health care practitioners may use this information to determine which treatment options are most likely to help their patient, and inform what to do to maximize the benefits from Vivitrol (e.g., try to ensure alcohol abstinence prior to Vivitrol administration and treat any comorbid alcohol use disorders) Overall, Vivitrol was found to work equally well across most of the subgroups with diverse characteristics.
- LIMITATIONS
-
- This was a sample of criminal justice involved individuals so the degree that the results generalize to non-criminal justice populations is unknown.
- About 33% of the patients randomized to treatment as usual received an opioid agonist medication. Many regions of the country do not have access to opioid agonist therapies, so practitioners must consider carefully if their patient would fare as well if referred to community resources based upon this study.
- There may be additional moderators or combinations of moderators that together affect the benefit of Vivitrol that were not studied here.
BOTTOM LINE
- For individuals & families seeking recovery: Extended release Vivitrol is a monthly injection that has been shown to reduce relapse in persons with opioid use disorder, if they make it through a potentially uncomfortable induction period of several days. This study found that if the patient was intoxicated in the past 30 days, the odds of relapse while using Vivitrol is about the same as if they were referred to community resources without getting Vivitrol. Unlike recent intoxication, many other patient characteristics did not modify or change the effect of Vivitrol, so overall it worked the same for almost everybody in the treatment group. If you or a loved one has an opioid use disorder Vivitrol can reduce relapse risk, especially when people are not drinking heavily.
- For scientists: This was secondary data analysis of a clinical trial comparing the effectiveness of Vivitrol to referral to community resources (i.e., treatment as usual) on opioid relapse. Moderator tests revealed that the medication worked equally well for all patient characteristics tested (e.g., age, gender, depression, suicidal thoughts, medical history, psychiatric history, family factors, employment, and others), however, patients with alcohol intoxication in the past 30 days performed no better than referral to treatment. Patients with recent alcohol intoxication might represent individuals who have substituted alcohol for opioids or represent greater complexity in the clinical picture that needs to be addressed.
- For policy makers: Vivitrol is an FDA approved medication for the treatment of opioid use disorder which can be useful for patients if they can make it through a potentially uncomfortable several day initial induction. This study found that Vivitrol patients who were intoxicated with alcohol in the past 30 days were no more or less likely to relapse than patients who were referred to community resources. All other patient characteristics tested (depression, suicidal thoughts, drug use risk, medial history, psychiatric history, employment, family issues, and others) appear to not matter in terms of treatment effectiveness to reduce relapse to opioid use. Many patients do not want to be on agonist therapy and Vivitrol may be a good alternative, especially if alcohol misuse and related disorders are addressed simultaneously.
- For treatment professionals and treatment systems: This study investigated if the effectiveness of Vivitrol was reduced among certain subgroups of patients compared to referral to community resources. The findings should be reassuring to practitioners given that except for the group that reported alcohol intoxication with in the past 30 days in treatment intake, the medication worked equally well across patients. Individuals with recent alcohol intoxication history may need additional treatment to address alcohol misuse to maximize the benefit Vivitrol provides for opioid outcomes.

