WHAT PROBLEM DOES THIS STUDY ADDRESS?
Cannabidiol, commonly known as CBD, is one of the primary psychoactive compounds commonly found in cannabis. It is approved by the Food and Drug Administration to treat rare and severe forms of disorders that cause epilepsy. However, due in part to medical marijuana advocacy, there has been a significant interest in exploring whether cannabidiol might have therapeutic value for other conditions. There is some evidence for generally modest effects on reductions in symptoms of some psychiatric conditions including substance use disorder, posttraumatic stress disorder, and anxiety disorder. However, no study has examined whether cannabidiol might reduce alcohol use. Research with rats demonstrated cannabidiol administration reduced alcohol intake over 7 days, and one human study found that cannabidiol reduced alcohol craving during a stress and alcohol cue exposure task. As such, cannabidiol may be helpful for reducing alcohol use itself in humans. This may be particularly true among people with posttraumatic stress disorder, where cannabidiol could help address anxiety-related symptoms of elevated arousal and avoidance that may lead to craving and higher levels of alcohol consumption. The current study examined safety and tolerability of cannabidiol and whether it might serve as a treatment for reducing alcohol and stress related symptoms within a sample of adults with alcohol use disorder, some of whom had a comorbid posttraumatic stress disorder diagnosis.
HOW WAS THIS STUDY CONDUCTED?
The current study included data on 54 participants across 2 randomized clinical trials (Study 1 n = 25; Study 2 n = 29) testing the tolerability and efficacy of cannabidiol relative to placebo in reducing alcohol craving and consumption. The trials were single-blinded, meaning that participants did not know whether they received cannabidiol or placebo, but some members of the research team did know. Also of note, the study did not measure whether participants could accurately guess if they received the study medication or not. This is sometimes done in studies where participants might easily guess if they have received the study medication (i.e., based on side effects) to account for expectations (see here for an example).
All participants had moderate or severe alcohol use disorder, at least 4 past month heavy drinking days (4+ drinks for women and 5+ for men), reported a desire to reduce alcohol consumption, and were willing to not use cannabis during the duration of the study. While this was verified by toxicology screen, many cannabidiol participants tested positive for THC during the study – the research team suggested that the study drug (and not illicit cannabis) was likely responsible for these positive tests. In Study 2, participants were also required to meet criteria for a DSM-5 diagnosis of posttraumatic stress disorder in addition to alcohol use disorder. In Study 1, participants randomized to the cannabidiol condition received 600 mg of cannabidiol per day for 4 weeks followed by 1200 mg of cannabidiol for 4 weeks. Participants in Study 2 received 600 mg per day for a total of 6 weeks. While groups in each study were found to be similar at baseline, in small samples like the ones here, it can be hard to detect baseline differences.
The researchers measured safety and tolerability by recording any adverse events, checking liver function throughout active medication administration, and by assessing for suicidality. Data on alcohol consumption was collected once per week in Study 1 and Study 2. Data on craving and symptoms of depression and anxiety were collected at baseline, 1-week, 4-week, 5-week, and 8-week follow up visits for Study 1. For Study 2, blood samples and data on craving and symptoms of depression, anxiety, and posttraumatic stress disorder were collected at baseline, 1-week, 4-week, and 6-week follow ups. Blood samples were also collected during these visits to determine the pharmacokinetics of cannabidiol, specifically the peak (highest concentration 45 minutes post-dose) and troughs (lowest concentration taken immediately prior to the next dose) of cannabidiol across the study period. The researchers reported on safety and tolerability, cannabidiol pharmacokinetics, and preliminary effect of cannabidiol on craving, alcohol consumption, and psychiatric symptoms relative to placebo. Finally, due to individual differences in cannabidiol pharmacokinetics, the researchers wanted to determine whether the effect of cannabidiol differed depending on cannabidiol levels in the blood.
WHAT DID THIS STUDY FIND?
There was no evidence that cannabidiol decreased alcohol consumption or other clinical outcomes
Across all study conditions, the average number of drinks per day and the percentage of heavy drinking days decreased across the study period. There were possibly some signals of small advantages for cannabidiol (see graphs below) in Study 1 (where participants had only alcohol use disorder) at some time points that were not statistically significant. The lack of significance may have been due in part, to the small study sample and that the sample was not drinking at a high intensity to begin with (e.g., only about 6 drinks per day on average). However, there was no effect of cannabidiol on alcohol use in Study 2 (where participants had co-occurring alcohol use and post-traumatic stress disorders), nor were there differences by treatment in anxiety or for posttraumatic stress symptoms (the latter of which was only measured in Study 2). The researchers did find an initial decrease for depressive symptoms; however, groups were similar on depressive symptoms as the study continued.
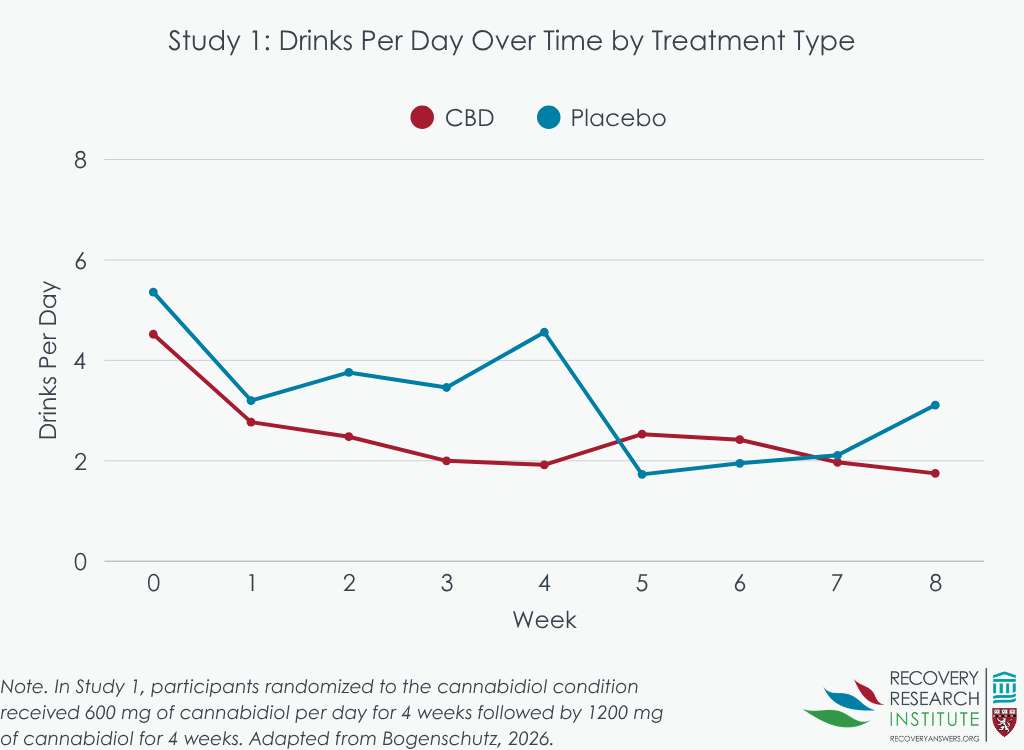
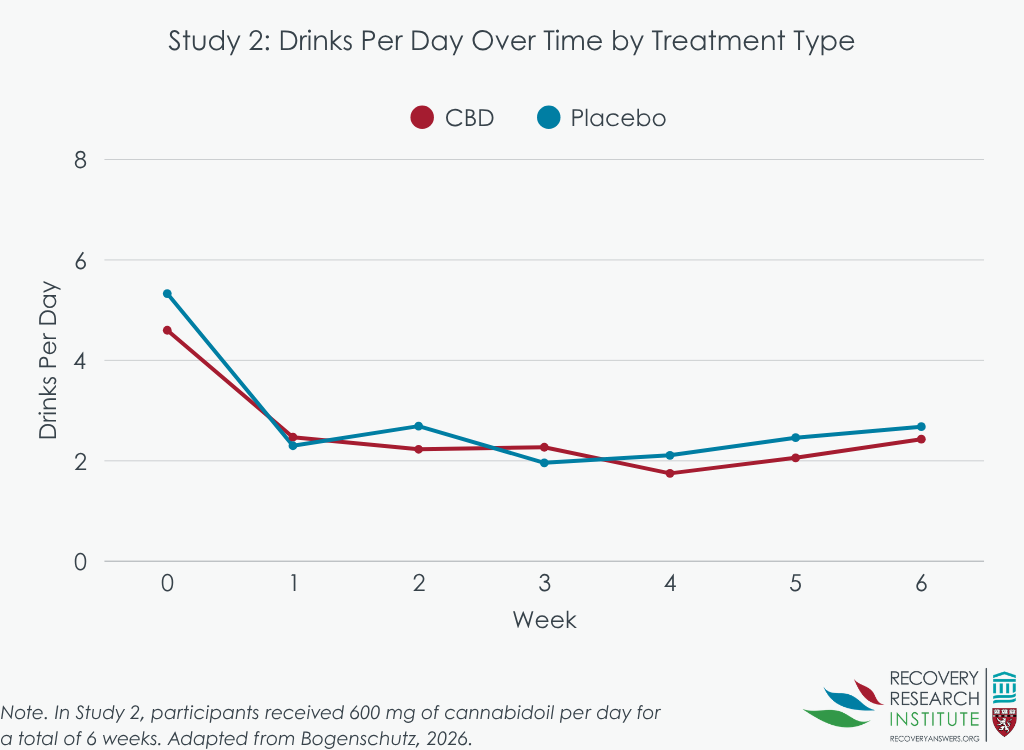
Cannabidiol levels increased over time and generally reached steady state within approximately 1 week
Across the first 4 weeks, troughs and peaks for participants in cannabidiol conditions across both studies increased at a similar rate. Statistical tests demonstrated that there were no differences from week 1 to week 4, indicating that steady state was generally achieved in the first week. In Study 1, dose was increased after week 4. For participants in Study 1, trough and peak continued to rise past week 4 and was significantly different from week 4 levels among participants in Study 2. There was variability in cannabidiol levels across participants, indicating individual differences in cannabidiol pharmacokinetics and metabolism.
Cannabidiol was generally well tolerated
Across both studies, nearly all adverse events were mild or moderate in severity. One patient randomized to cannabidiol experienced a case of diabetic ketoacidosis 25 days after the last dose of cannabidiol. This experience was judged to be possibly related to the medication, because cannabis use is known to increase the risk of ketoacidosis in patients with type 1 diabetes. Otherwise, the most common adverse events in the cannabidiol group included diarrhea (27%), somnolence (23%), and nausea (20%), though only nausea occurred significantly more in the cannabidiol group relative to placebo. Suicidal ideation occurred once in each treatment group across both studies, and elevated liver enzymes were observed among 2 participants randomized to cannabidiol, and 1 randomized to placebo. Seven study participants randomized to cannabidiol stopped, and 3 reduced their study medication due to adverse events.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?
These results suggest that cannabidiol might be tolerable and relatively safe. Though the sample size was small, and therefore not powered statistically speaking to detect differences, the actual size of the observed average differences between the active medication and the placebo controlled groups suggest that using cannabidiol may not reduce alcohol consumption or clinical symptoms of depression, anxiety, or posttraumatic stress disorder among people with alcohol use disorder such as those participating in this study. Notably, the studies should be interpreted in context of the small study samples, which can make it challenging to create equivalent medication and placebo groups, and the lack of test for whether participants could accurately guess whether they received the study medication. These nuances could affect the results in unknown ways.
Nevertheless, these studies are important because there has been a lot of increased interest in the potential therapeutic effect of cannabidiol, and these data do not support this expectation. The lack of effect may be, in part, due to a lower level of absorbed cannabidiol than is necessary to detect a therapeutic effect on alcohol craving, at least according to recent lab studies. Alternatively, cannabidiol may be more effective among other populations, such as those with more mild symptoms of alcohol use disorder. Future research will need to evaluate these hypotheses. However, for now, those looking for new tools to help reduce alcohol consumption or psychiatric concerns such as depression, anxiety, or posttraumatic stress disorder among those with alcohol use disorder should know that cannabidiol may be unlikely to provide clinical benefit, and may lead to increased nausea.
BOTTOM LINE
Cannabidiol, a common psychoactive element in cannabis, was generally well tolerated and safe in this study, but appeared to produce some minor side effects like nausea and a small potential risk for more serious adverse events. However, there was no evidence that cannabidiol might be helpful at reducing alcohol consumption or craving, or symptoms of anxiety, depression, and posttraumatic stress disorder in this particular sample with alcohol use disorder, compared to a placebo.
- For individuals and families seeking recovery: If you or a loved one is interested in making a change to your drinking, cannabidiol may not help reduce your drinking or other symptoms like depression and anxiety, and it may lead to nausea. There are effective medications for alcohol use that are approved by the Food and Drug Administration, such as naltrexone, acamprosate, and disulfiram. There are also many other options that have been shown to be effective at reducing alcohol use, including psychosocial treatments (12-step facilitation, cognitive behavioral therapy, relapse prevention, etc.).
- For treatment professionals and treatment systems: Many patients may be curious about cannabidiol as a treatment for their mental health concerns, including alcohol use disorder, depression, anxiety, and posttraumatic stress disorder. This particular study suggests that cannabidiol may not be helpful for these conditions.
- For scientists: These findings do not provide support for the efficacy of cannabidiol at decreasing alcohol consumption or symptoms of depression, anxiety, and posttraumatic stress disorder in this sample. However, more work with different and larger sample sizes is needed to confirm. Other research suggests that a higher dose than that provided in this study may be necessary to achieve a therapeutic effect. Alternatively, cannabidiol may only be helpful for certain groups or people. Future research may determine whether cannabidiol could be useful for some subgroups.
- For policy makers: Though cannabidiol is currently legal, it is prohibited to add cannabidiol to any food product or to sell it as a dietary supplement. The results of this research do not provide evidence for any therapeutic benefit, though it suggests that cannabidiol is generally well tolerated. More evidence would be needed to change its status, and in general, people seeking care for alcohol use should be encouraged at a public health level to consider finding treatments supported by research.
CITATIONS
Bogenschutz, M. P., Blessing, E., Dgheim, D., Cho, D., Zhang, J., Laska, E. M., & Marmar, C. R. (2026). Effects of cannabidiol in alcohol use disorder patients with and without co‐occurring post‐traumatic stress disorder: Tolerability but no evidence for efficacy in two randomized proof‐of‐concept trials. Alcohol: Clinical and Experimental Research, 50(1). doi: 10.1111/acer.70212.














