WHAT PROBLEM DOES THIS STUDY ADDRESS?
Buprenorphine/naloxone is a medication that reduces opioid use and opioid-related overdose risk. However, many people face barriers to accessing this life-saving treatment. Traditional buprenorphine/naloxone induction typically requires patients visit a clinic daily for the first 3 days to receive supervised doses and have their symptoms monitored. These frequent visits can create work, family, and transportation challenges. These obstacles disproportionately affect people living in rural areas and those with limited resources. While telemedicine offers a potential solution to expand access to buprenorphine/naloxone treatment, most prior research has been observational rather than experimental. This study addressed the need for rigorous evidence comparing telemedicine-assisted buprenorphine/naloxone induction with traditional in-person care. The researchers tested whether combining an initial in-person visit with telemedicine follow-ups would perform as well as standard care, while reducing barriers to buprenorphine/naloxone access.
HOW WAS THIS STUDY CONDUCTED?
This randomized controlled trial testing telemedicine-assisted buprenorphine/naloxone induction enrolled 138 adults diagnosed with opioid use disorder with access to a cell phone and a responsible adult family member. Participants were recruited from an addiction treatment center in India between December 2023 and August 2024 and randomly assigned to receive either standard buprenorphine/naloxone induction or telemedicine-assisted buprenorphine/naloxone induction. Those in the standard induction group attended in-person clinic visits for 3 consecutive days to receive supervised buprenorphine/naloxone doses and clinical monitoring. Those in the telemedicine induction group received their first dose in person on day 1, and a responsible adult was trained to administer medication and monitor symptoms. These participants then had daily telephone or video consultations with the clinical team over days 2 through 6. Both groups returned for an in-person evaluation on day 7 and continued receiving buprenorphine/naloxone treatment as routine care afterward. See graphic below for an overview.
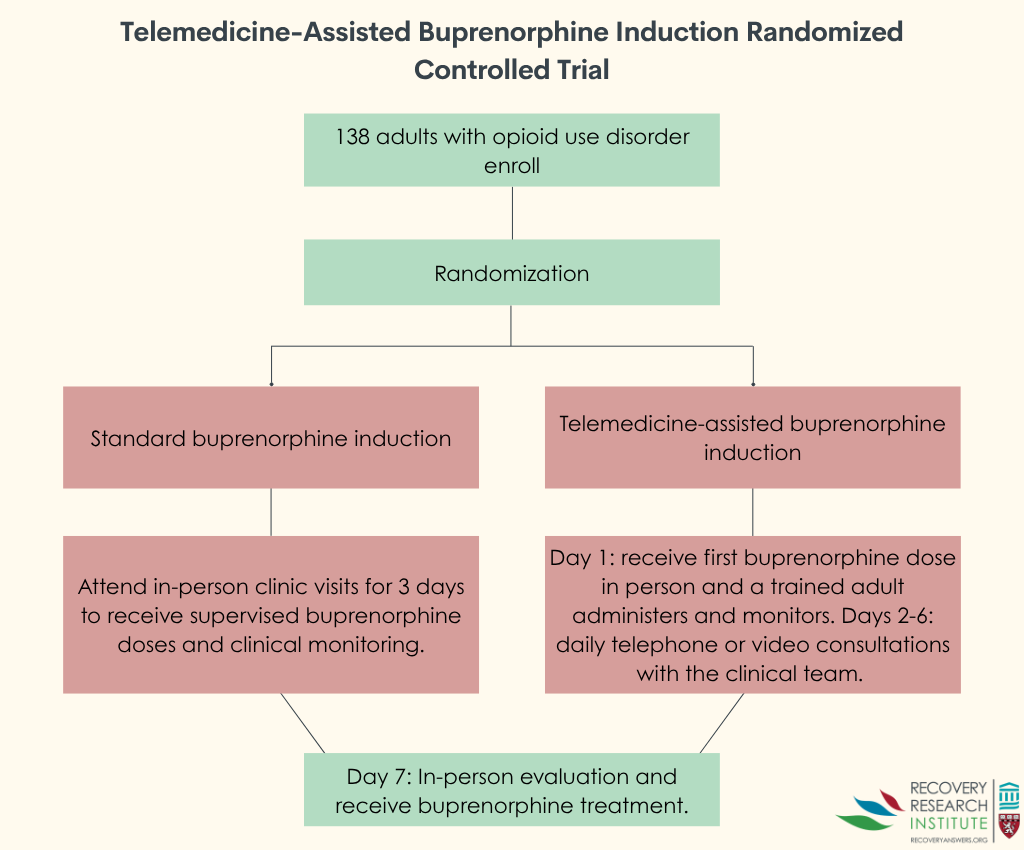
The researchers followed participants for 1 month, measuring whether they stayed in treatment and tracking outcomes such as abstinence from illicit opioids, withdrawal symptoms, cravings, quality of life, patient satisfaction, and medication adherence through self-reports and urine drug screens. Because this study focused primarily on treatment retention during the critical first week, attendance at the in-person visit on or around day 7 was used as the main indicator of whether participants had stayed engaged in care. The trial tested for non-inferiority with the researchers hypothesizing that telemedicine would do just as well as standard care. The study sample was on average 28 years old and mostly male (98.5%). While the study states that the two groups were equivalent at baseline, there were no statistical tests reported examining group differences. This could have unknown effects on study outcomes.
WHAT DID THIS STUDY FIND?
Retention rates were comparable between groups
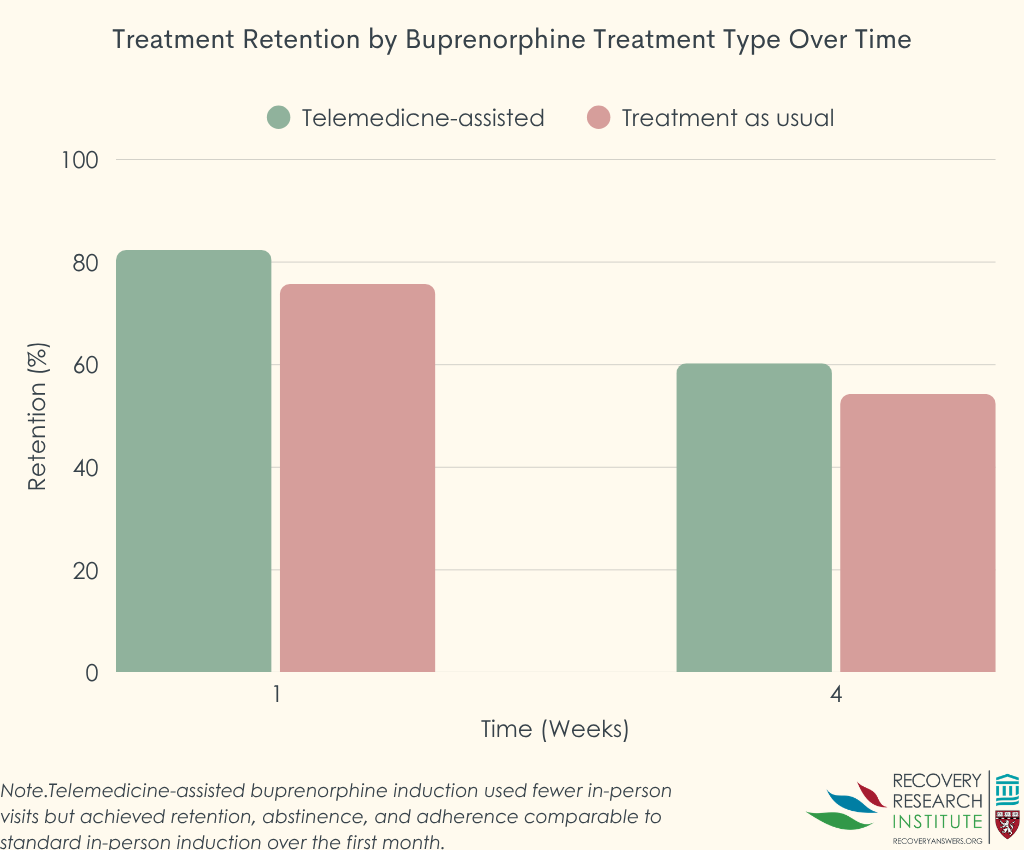
Among those receiving telemedicine-assisted induction, 82% of people stayed in treatment through the first week compared with 76% in the standard care group. At 1 month, about 60% of people in the telemedicine group and 54% in the standard care group remained in treatment. See graph below.
Clinical outcomes improved similarly in both groups
Both treatment approaches led to substantial reductions in withdrawal symptoms and cravings. Withdrawal symptoms decreased by about 70% in both groups during the first week. Quality of life improved by roughly 30% across all areas measured, including physical health, psychological well-being, social relationships, and environment. These improvements continued through the 1-month follow-up period.
Patient experiences were generally positive with both models
People in both groups reported high satisfaction with their treatment and felt their providers showed empathy and understanding. Patient satisfaction was similar in both groups, exceeding 80% of the maximum possible score threshold. About 90% of participants in both groups took their medication as prescribed during the first week. Abstinence from illicit opioids was similar between groups, with roughly 80% of participants in each group reporting no illicit opioid use at the 1-week mark. Known side effects such as mild sedation and constipation occurred at similarly low rates in both groups, and no serious safety concerns emerged.
Study strengths and limitations
This study provided rigorous experimental evidence through random assignment and tracked multiple important outcomes beyond just staying in treatment. However, about one-quarter of participants in each group dropped out before the 1-week follow-up, making longer-term comparisons less certain. In addition, the research took place at a single treatment center, and the sample was comprised of mostly young men, which may limit how well these findings generalize to other settings and populations. The study did not assess psychiatric conditions using structured diagnostic tools, so rates of mental health problems may have been underestimated. The study also did not examine cost differences between the two approaches. Also, of note, was that overall, by week 4, approximately 40-50% of patients in both groups had discontinued their buprenorphine/naloxone treatment indicating the challenge of keeping patients on these medications regardless of whether clinics use telemedicine or in-person approaches.
WHAT ARE THE IMPLICATIONS OF THE STUDY FINDINGS?
This study’s findings with a sample of mostly young men in India, align with previous observational studies indicating telemedicine-assisted induction may be a viable option for people starting buprenorphine/naloxone for opioid use disorder. The hybrid model tested here, which combined an initial in-person visit with remote follow-up care, achieved results comparable to traditional approaches that require multiple consecutive days of clinic visits. Telemedicine reduces many barriers to care and may be particularly valuable for people living in rural areas or those facing transportation challenges, work conflicts, or concerns about stigma associated with visiting addiction treatment centers. The success of this hybrid model could inform policy discussions about appropriate regulatory frameworks that balance safety with accessibility. It is worth noting, however, that 55-60% of patients remained in buprenorphine/naloxone treatment by 1 month across both groups; drop-out from opioid use disorder medication treatment remains a challenge. More research is needed to understand how telemedicine-assisted induction works in diverse populations, including among women and people with co-occurring mental health conditions, and whether it remains helpful over longer time periods. Cost-effectiveness studies would also help healthcare systems make informed decisions about implementing such programs.


BOTTOM LINE
Telemedicine-assisted buprenorphine/naloxone induction, which combines an initial in-person visit with remote follow-up care, kept people in treatment just as well as traditional approaches requiring multiple consecutive clinic visits. This hybrid model offers a further treatment modality option that may reduce barriers to accessing life-saving medication treatment for opioid use disorder.
- For individuals and families seeking recovery: People may be able to start buprenorphine/naloxone treatment for opioid use disorder in person and then check in by phone or video, with similar short-term outcomes to daily in-person visits. Availability of this option will vary by location, depending on local regulations and clinic policies, so individuals should ask their treatment providers what is offered in their area.
- For treatment professionals and treatment systems: This trial provides some further evidence that telemedicine-assisted buprenorphine/naloxone induction may do as well as traditional in-person induction in terms of treatment retention and clinical outcomes. Adoption of this model may help programs expand capacity, reach underserved populations, and reduce patient burden without necessarily sacrificing treatment quality and safety.
- For scientists: This randomized controlled non-inferiority trial demonstrated that telemedicine-assisted buprenorphine/naloxone induction met non-inferiority criteria compared with standard care for 1-week retention. Future research should examine implementation with more diverse demographic and clinical samples and settings, longer follow-up periods, conduct cost-effectiveness analyses, and examine patients with varying degrees of psychiatric comorbidity, as well as investigate fully remote induction models.
- For policy makers: This randomized trial from India found that telemedicine-assisted buprenorphine/naloxone induction achieved retention and clinical outcomes comparable to traditional supervised in-person dosing while potentially reducing treatment barriers. These findings could inform regulatory frameworks that maintain safety while expanding access to buprenorphine/naloxone treatment for opioid use disorder.
CITATIONS
Dhillon, H. S., Ghosh, A., Naik, S., Nanjayya, S., Verma, A., & Basu, D. (2025). Telemedicine-assisted buprenorphine induction versus standard of care: A randomized controlled non-inferiority trial. Addiction, 121(4), 883-893. doi: 10.1111/add.70269.
 Starting buprenorphine/naloxone medication for opioid use disorder often requires multiple clinic visits during the first week, which can be a barrier for some. This study tested whether a hybrid approach using telemedicine for follow-up visits after an initial in-person appointment worked as well as the traditional model requiring 3 consecutive days of in-person visits.
Starting buprenorphine/naloxone medication for opioid use disorder often requires multiple clinic visits during the first week, which can be a barrier for some. This study tested whether a hybrid approach using telemedicine for follow-up visits after an initial in-person appointment worked as well as the traditional model requiring 3 consecutive days of in-person visits.














